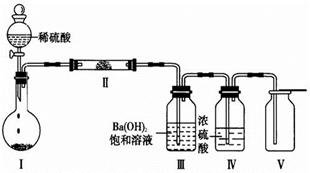
��Ŀ����
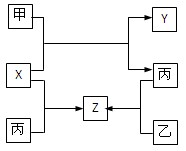
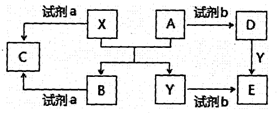
(15��)A��B��C��D��Ϊ��ѧ��ѧ�ij��������Ҿ�����ͬһ��Ԫ�أ�����֮���ת����ϵ����ͼ��ʾ(��Ӧ���������������Ѿ���ȥ)��
A B
B C
C D
D
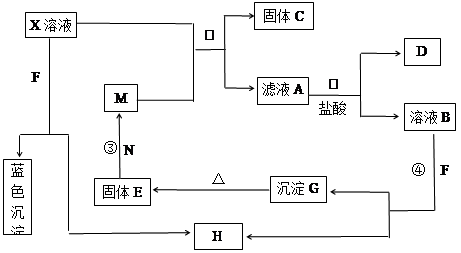
(1)��A��D��ˮ��Һ����ʹʪ�����ɫʯ����ֽ��죬��A��D�ֱ�Ϊ(��д��ѧʽ)��
A ��D ��
(2)��A��ˮ��Һ��ʹʪ��ĺ�ɫʯ����ֽ������D��ϡ��Һ��ʹ��ɫ��ʪ��ʯ����ֽ��죬��AΪ ��д��A Bת���Ļ�ѧ����ʽ�� ��
Bת���Ļ�ѧ����ʽ�� ��
(3)��AΪ���ý���Ԫ�صĵ��ʣ�DΪǿ���ɫ��Ӧ�Ի�ɫ����A�� ��
B�� ��C�� ��C����Ϊ������ߵ�������C�������̼������Ӧ�Ļ�ѧ����ʽΪ�� ��A��ˮ������Ӧ�Ļ�ѧ����ʽ ��
(4)��AΪ�����ķǽ������ʣ���A�ж������������д�±����������Ӧ�Ļ�ѧ��Ӧ����ʽ��(���Բ�������Ҳ���Բ���)
A
 B
B C
C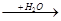 D
D(1)��A��D��ˮ��Һ����ʹʪ�����ɫʯ����ֽ��죬��A��D�ֱ�Ϊ(��д��ѧʽ)��
A ��D ��
(2)��A��ˮ��Һ��ʹʪ��ĺ�ɫʯ����ֽ������D��ϡ��Һ��ʹ��ɫ��ʪ��ʯ����ֽ��죬��AΪ ��д��A
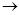 Bת���Ļ�ѧ����ʽ�� ��
Bת���Ļ�ѧ����ʽ�� ��(3)��AΪ���ý���Ԫ�صĵ��ʣ�DΪǿ���ɫ��Ӧ�Ի�ɫ����A�� ��
B�� ��C�� ��C����Ϊ������ߵ�������C�������̼������Ӧ�Ļ�ѧ����ʽΪ�� ��A��ˮ������Ӧ�Ļ�ѧ����ʽ ��
(4)��AΪ�����ķǽ������ʣ���A�ж������������д�±����������Ӧ�Ļ�ѧ��Ӧ����ʽ��(���Բ�������Ҳ���Բ���)
| ��� | A | �� B��C��ѧ����ʽ |
| �� | | |
| �� | | |
| �� | | |
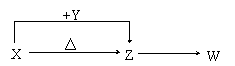
����A B
B C
C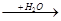 Dת����ϵ����ϣ�
Dת����ϵ����ϣ�
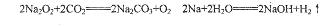
S SO2
SO2 SO3
SO3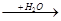 H2SO4 ��
H2SO4 ��
H2S SO2
SO2 SO3
SO3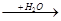 H2SO4 ��
H2SO4 ��
N NO
NO NO2
NO2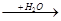 HNO3 ��
HNO3 ��
C CO
CO CO2
CO2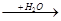 H2CO3 ��
H2CO3 ��
NH3 NO
NO NO2
NO2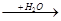 HNO3 �ȣ�
HNO3 �ȣ�
(1)ˮ��Һ����ʹʪ�����ɫʯ����ֽ��죬��Һ�����ԣ���
H2S SO2
SO2 SO3
SO3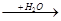 H2SO4 ��
H2SO4 ��
(2)��ʹʪ��ĺ�ɫʯ����ֽ��������Һ�Լ��ԣ���
NH3 NO
NO NO2
NO2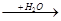 HNO3 ��
HNO3 ��
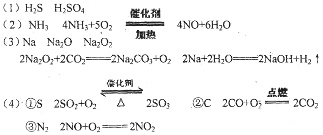
(3)��ɫ��Ӧ�Ի�ɫ������AΪNa��Na Na2O
Na2O Na2O2
Na2O2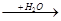 NaOH�����漰�ķ�Ӧ��
NaOH�����漰�ķ�Ӧ��
 B
B C
C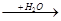 Dת����ϵ����ϣ�
Dת����ϵ����ϣ� 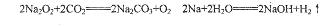
S
 SO2
SO2 SO3
SO3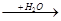 H2SO4 ��
H2SO4 ��H2S
 SO2
SO2 SO3
SO3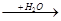 H2SO4 ��
H2SO4 ��N
 NO
NO NO2
NO2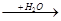 HNO3 ��
HNO3 ��C
 CO
CO CO2
CO2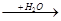 H2CO3 ��
H2CO3 ��NH3
 NO
NO NO2
NO2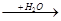 HNO3 �ȣ�
HNO3 �ȣ�(1)ˮ��Һ����ʹʪ�����ɫʯ����ֽ��죬��Һ�����ԣ���
H2S
 SO2
SO2 SO3
SO3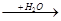 H2SO4 ��
H2SO4 ��(2)��ʹʪ��ĺ�ɫʯ����ֽ��������Һ�Լ��ԣ���
NH3
 NO
NO NO2
NO2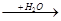 HNO3 ��
HNO3 ��(3)��ɫ��Ӧ�Ի�ɫ������AΪNa��Na
 Na2O
Na2O Na2O2
Na2O2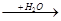 NaOH�����漰�ķ�Ӧ��
NaOH�����漰�ķ�Ӧ��
��ϰ��ϵ�д�
�����Ŀ