��Ŀ����
��ѹǿΪ2��20��104 kPa���¶ȴﵽ374��ʱ��ˮ��Ϊ�����ٽ�״̬������ʱˮ�ɽ�CO2�Ⱥ�̼������ת��Ϊ�л������ǡ�ˮ�ȷ�Ӧ�����������ڵ��¸��¸�ѹ������ͨ��ˮ�ȷ�Ӧ������ʯ�͡�ú�ȿ�����Դ������˵������ȷ���ǣ� ��
| A��������̼�볬�ٽ�ˮ�����������͵ķ�Ӧ�����ڷ��ȷ�Ӧ |
| B����ˮ�ȷ�Ӧ����һ�ָ��ӵ�������ѧ�仯 |
| C���������糧�������÷��ȣ���������̼ת��Ϊ��Դ���� |
| D�����ſƼ��Ľ�������ˮ�ȷ�Ӧ����ȡ��Դ����ʵ�ֵ�����̼��Դ�ĺ�гѭ�� |
A
�������������A�����¸�ѹ������ͨ��ˮ�ȷ�Ӧ������ʯ�͡�ú�ȿ�����Դ���������ȷ�Ӧ��A���� B��ѹǿ�ﵽ2��20��104kPa���¶ȴﵽ374��ʱ��ˮ��Ϊ�����ٽ�״̬�����ù���Ϊ�����仯����ʱˮ�ɽ�CO2 �Ⱥ�̼������ת��Ϊ�л��Ϊ��ѧ�仯����B��ȷ�� C����ѹ�����½�CO2�Ⱥ�̼������ת��Ϊ�л���ǽ�������̼ת��Ϊ��Դ���ʣ���C��ȷ��D��������õ����ϵ�̼��Դ��������̼��Դ��ѭ��ʹ�ã���D��ȷ��
���㣺��������Դ�Ŀ���������

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д���֪����CO��g����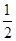 O2��g��=CO2��g������H����283.0 kJ��mol��1
O2��g��=CO2��g������H����283.0 kJ��mol��1
��H2��g����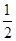 O2��g��=H2O��g�� ��H����241.8 kJ��mol��1
O2��g��=H2O��g�� ��H����241.8 kJ��mol��1
��CO��g����H2O��g��=H2��g����CO2��g���Ħ�HΪ
| A��+41.2 kJ?mol-1 | B����41.2 kJ?mol-1 | C��+82.4kJ?mol-1 | D����524.8 kJ?mol-1 |
���й��ڷ�Ӧ�����������仯��˵����ȷ���� �� ��
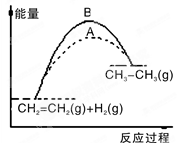
| A��ͼ��a��b���߿ɷֱ��ʾ��ӦCH2��CH2(g)��H2(g)��CH3CH3(g)��H��0ʹ�ú�δʹ�ô���ʱ����Ӧ�����е������仯 |
| B����֪2C(s)��2O2(g)��2CO2(g) ��H1��2C(s)��O2(g)��2CO(g) ��H2����H1����H2 |
| C��ͬ��ͬѹ�£���ӦH2(g)��Cl2(g)��2HCl(g)�ڹ��պ͵�ȼ�����µĦ�H��ͬ |
| D����һ�������£�ij���淴Ӧ�Ħ�H����100kJ��mol��1����÷�Ӧ����Ӧ��ܱ��淴Ӧ��ܴ�100kJ��mol��1 |
ָ�����£�1 mol��ѧ���ֽ����̬ԭ������Ҫ��������E��ʾ����ϱ�����Ϣ�ж�����˵������ȷ����
| ���ۼ� | H��H | F��F | H��F | H��Cl | H��I |
| E(kJ��mol-1) | 436 | 157 | 568 | 432 | 298 |
A��432 kJ��mol-1��E(H��Br)��298 kJ��mol-1
B���������ȶ��Ĺ��ۼ���H��F��
C��H2(g)��2H (g) ?H����436 kJ��mol-1
D��H2(g)��F2(g)��2HF(g) ?H����25 kJ��mol-1
����ͼ��ֱ��ʾ�йط�Ӧ�ķ�Ӧ�����������仯�Ĺ�ϵ��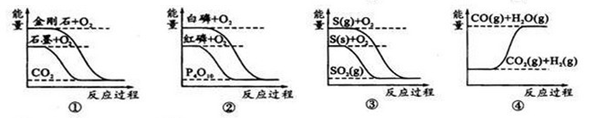
�ݴ��ж�����˵����ȷ����
| A��ʯīת��Ϊ���ʯ�����ȷ�Ӧ |
| B�����ױȺ����ȶ� |
| C��CO(g)+H2O(g)=CO2(g)+H2(g)��H��0 |
| D��S(g)+O2(g)=SO2(g) ��H1�� S(s)+O2(g)=SO2(g) ��H2����H1����H2 |
�����й��Ȼ�ѧ����ʽ��������ȷ����(�� ��)
| A����ϡ��Һ�У�H��(aq)����OH��(aq) = H2O(l)����H����57.3 kJ/mol��������0.6 mol H2SO4��ϡ�����뺬1 mol NaOH����Һ��ϣ��ų�����������57.3 kJ |
| B����֪C(ʯī��s)=C(���ʯ��s)����H��0������ʯ��ʯī�ȶ� |
| C����Ҫ���ȵķ�Ӧ˵���������ȷ�Ӧ |
| D����֪2C(s)��2O2(g)=2CO2(g)����H1��2C(s)��O2(g)=2CO(g)����H2����H1����H2 |
�к��Ȳⶨʵ���У���50mL0.50mol/L�����50mL0.55mol/LNaOH����ʵ�飬����˵��������ǣ� ��
| A������60mL 0.50mol/L�����50mL 0.55 mol/L NaOH��Һ���з�Ӧ��������к�����ֵ��ԭ����ͬ |
| B����50mL0.50mol/L�����50mL0.55mol/LNaOH����ʵ�����50mL0.50mol/L����50mL0.50mol/LNaOH��õ���ֵȷ |
| C�������ʱ����Ͳ��NaOH��ҺӦ��������С�ձ��У������ò��������� |
| D��װ���еĴ�С�ձ�֮����������ĭ���ϵ������DZ��¸��ȡ�����������ʧ |