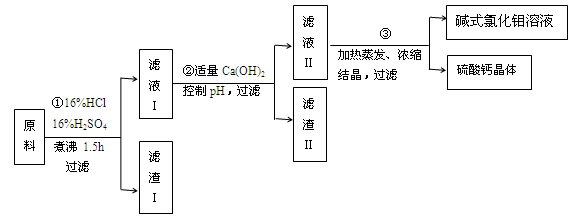
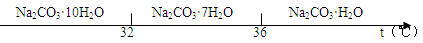ЬтФПФкШн
ЯТСагаЙиЮяжЪМьбщЕФЪЕбщНсТле§ШЗЕФЪЧЃЈ ЃЉ
| бЁЯю | ЪЕбщВйзїМАЯжЯѓ | ЪЕбщНсТл |
| A | ЯђФГШмвКжаМгШыЯѕЫсЫсЛЏЕФТШЛЏБЕШмвКЃЌгаАзЩЋГСЕэЩњГЩ | ИУШмвКжавЛЖЈКЌгаSO |
| B | ЯђФГШмвКжаМгШы2ЕЮKSCNШмвКЃЌШмвКВЛЯдбЊКьЩЋЁЃдйЯђШмвКжаМгШыМИЕЮаТжЦЕФТШЫЎЃЌШмвКБфЮЊбЊКьЩЋ | ИУШмвКжавЛЖЈКЌгаFe2ЃЋ |
| C | ФГЦјЬхФмЪЙЪЊШѓЕФРЖЩЋЪЏШяЪджНБфКь | ИУЦјЬхЫЎШмвКвЛЖЈЯдМюад |
| D | НЋЩйСПФГгаЛњЮяЕЮМгЕНаТжЦЧтбѕЛЏЭаќзЧвКжаЃЌГфЗжеёЕДЃЌЮДМћзЉКьЩЋГСЕэЩњГЩ | ИУгаЛњЮяВЛКЌШЉЛљ |
B
AЯюЃЌAgЃЋЁЂSOОљИЩШХЃЛCЯюЃЌЪЙРЖЩЋЪЏШяЪджНБфКьЃЌгІГЪЫсадЃЛDЯюЃЌВйзїЪБгІМгШШЕНЗаЬкЁЃ

СЗЯАВсЯЕСаД№АИ
 УћаЃПЮЬУЯЕСаД№АИ
УћаЃПЮЬУЯЕСаД№АИ
ЯрЙиЬтФП

 ЁЂSO
ЁЂSO РызгЕФЪЕбщЗНЗЈе§ШЗЕФЪЧ________(ЬюБрКХ)ЁЃ
РызгЕФЪЕбщЗНЗЈе§ШЗЕФЪЧ________(ЬюБрКХ)ЁЃ
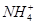 ЁЂKЃЋЁЂNaЃЋЁЂMg2ЃЋЁЂBa2ЃЋЁЂAl3ЃЋЁЂClЃЁЂIЃЁЂ
ЁЂKЃЋЁЂNaЃЋЁЂMg2ЃЋЁЂBa2ЃЋЁЂAl3ЃЋЁЂClЃЁЂIЃЁЂ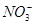 ЁЂ
ЁЂ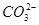 ЁЂS2ЃЁЂ
ЁЂS2ЃЁЂ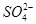 ЁЂ
ЁЂ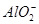 ЁЂ
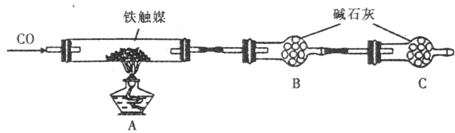
ЁЂ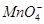 ЃЌШЁИУШмвКНјаавдЯТЪЕбщЃК
ЃЌШЁИУШмвКНјаавдЯТЪЕбщЃК