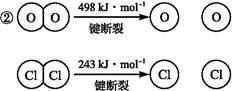
��Ŀ����
����A��B��C��D��E��F���ֳ����������֪���ǰ�������������K����Ag����Na����
Ba2����Fe2����Al3������������Cl����OH����AlO2-��NO3-��SO42-��CO32-�������Ƿֱ����0.1 mol/L����Һ��������ʵ�飺
�ٲ����ҺA��C��E���ʼ��ԣ��Ҽ���A��E��C��E����ɫ��dz��ɫ������ɫ�ܲ����۲죩������B��Һ�еμ�ϡ��ˮ�������������ɳ����������ȫ���ܽ⣻����F��Һ�еμ�ϡ���ᣬ��Һ����ػ�ɫ��������ɫ�������ɣ�����D��Һ�еμ�Ba��NO3��2��Һ����������
��1��д��A��D��E��F�Ļ�ѧʽ��
A________��D________��E________��F________��
��2�������ӷ���ʽ����C��Һ�ʼ��Ե�ԭ��__________________________________��
��3��д��ʵ����з�Ӧ�����ӷ���ʽ��________________________________��
��1��Ba��OH��2 AlCl3 KAlO2 FeSO4
��2��CO32-��H2O HCO3-��OH��
HCO3-��OH��
��3��3Fe2����NO3-��4H��=3Fe3����NO����2H2O
����

�ɼ������ӻ�������ɵĻ����������������е������֣�K����NH��Ba2����Al3����Fe3����Cl����SO42-��CO32-�����û��������ˮ��ó�����Һ����ȡ3��100 mL����Һ�ֱ��������ʵ�飺
| ��� | ʵ������ | ʵ���� |
| 1 | �ӹ������� | ��������� |
| 2 | ������NaOH��Һ������ | �ռ�������1.12 L(������ɱ�״���µ����)�����к��ɫ�������ɣ�����������ϴ�ӡ�������գ����ص�1.60 g���塣 |
| 3 | ������BaCl2��Һʱ�������ó�������ϴ�ӡ�������� | ��һ�γ�������Ϊ2.33 g |
�Իش��������⣺
(1)����ʵ��1��CO32-�Ƿ���ڵ��ж���________��(�һ�����ڡ���һ�������ڡ�����ȷ����)������ʵ��1��3�жϻ������һ�������ڵ�������________��
(2)��ȷ����Һ��һ�����ڵ������Ӽ������ʵ���Ũ��(�ɲ�����)��
| �����ӷ��� | ���ʵ���Ũ��(mol��L��1) |
| | |
| | |
| | |
A��B��C��D��E��F��G�������ʼ������ͼ��ʾ��ת����ϵ������A��B��D��G����ͬ��Ԫ�ء�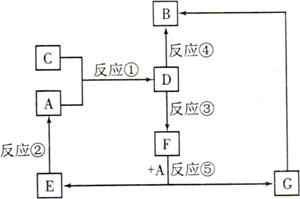
��֪��
AΪ�������ʣ�BΪ���ɫ���壬EΪ�ܶ���С�����壬GΪdz��ɫ����Һ��
D��ˮ��ҺΪ��ɫ��Һ��������������Һ��Ӧ���ɲ�����ϡ����İ�ɫ������
��ˮ��Һ��D�ܽ�ij����������ΪF��F�Ǻ�������Ԫ�صĻ����
��ش��������⣺
��1������C���ʵ�Ԫ�������ڱ��е�λ���� ���ڶ���������Ԫ���У���Ԫ����������Ԫ�ص�ԭ�Ӱ뾶�Ӵ�С��˳���� (��Ԫ�ط��ű�ʾ)��
��2��D��ˮ��Һ�� �ԣ��������ӷ���ʽ����ԭ�� ��
��3��������Ӧ�������û���Ӧ���� (�����)��
��4����Ӧ��(��D��ij������������ΪF)�����ӷ���ʽ�� ��
��5��������C��������ʵ�顣��֪������Ӧ�����У�ÿ����0.1mol KI��ת�Ƶĵ�����ԼΪ3.612��1023�����밴��Ҫ����գ�
| ʵ�鲽�� | ʵ������ | д���ӷ���ʽ |
| ����������ͨ�����KI��Һ | ��Һ������ ɫ | |
| ����ͨ������ | ��Һ�����ɫ | |
ij��ҵ��ˮ�����±��е�ijЩ���ӣ��Ҹ������ӵ����ʵ���Ũ����ȣ���Ϊ0.1 mol/L(����ֵ����ˮ�ĵ��뼰���ӵ�ˮ��)��
| ������ | K����Ag����Mg2����Cu2����Al3����NH4+ |
| ������ | Cl����CO32����NO3����SO42����I�� |
��ͬѧ��̽����ˮ����ɣ�����������ʵ�飺
��.ȡ����ɫ��Һ5 mL���μ�һ�ΰ�ˮ�г������ɣ��������������ӡ�
��.�ò�˿պȡ��Һ���ڻ��������գ�����ɫ�ܲ����۲죬����ɫ���档
��.��ȡ��Һ����������ᣬ����ɫ�������ɣ�����ɫ������������ɺ���ɫ��
��.��������õ���Һ�м���BaCl2��Һ���а�ɫ�������ɡ�
���ƶϣ�
(1)�ɢ��жϣ���Һ��һ�������е���������___________________________________��
(2)���м�������������ɫ��������ӷ���ʽ��________________________��
(3)��ͬѧ����ȷ��ԭ��Һ��������������________����������________�����ݴ��Ʋ�ԭ��ҺӦ�ó�________�ԣ�ԭ����____________________________(�������ӷ���ʽ˵��)��
(4)��ȡ100 mLԭ��Һ������������NaOH��Һ���˹������漰�����ӷ���ʽΪ____________________________����ַ�Ӧ����ˣ�ϴ�ӣ����ճ��������أ��õ��Ĺ�������Ϊ________g��
��.ͭ�����仯�������ճ�������Ӧ�ù㷺��ij�о���ѧϰС���ô�ͭ��������Fe�������������Ӧ�ù���A����ϡ�����ܽ�A��Ȼ����Լ�������Һ��pH�����ҺB����ҺB��ϵ�в����ɵ��Ȼ�ͭ���壬��ش�
��1������A��ϡ�����ܽ��ԭ���� __��
��2��������ҺB���Ƿ����Fe3���ķ����� __��
��3����֪Ԫ���ڸ�̬ʱ�����������ԣ���������CuSO4��Һ�м���һ������Na2SO3��NaCl��Һ�����ȣ�����CuCl������������CuCl�����ӷ���ʽ�� ��
��.�����£�ijͬѧ��ϡ����Ͱ�ˮ�������ϣ�������Һ��Ũ�Ⱥͻ�Ϻ�������Һ��pH���±���
| ʵ���� | ��ˮ���ʵ���Ũ��/��mol��L��1�� | �������ʵ���Ũ��/��mol��L��1�� | �����ҺpH |
| �� | 0.1 | 0.1 | pH��5 |
| �� | c | 0.2 | pH��7 |
| �� | 0.2 | 0.1 | pH>7 |
��ش�
��4���ӵڢ�������������������û����Һ����ˮ�������c��H������ __mol��L��1���ӵڢ������������c __���>����<��������0.2 mol��L��1���ӵڢ������������֪�������Һ��c��NH4+�� __���>����<��������c��NH3��H2O����
��5��д������������ҺNH4+Ũ���ɴ�С��˳�� __> __> __> __����ѡ���ţ���
A��0.1 mol��L��1 NH4Cl
B��0.1 mol��L��1 NH4Cl��0.1 mol��L��1 NH3��H2O
C��0.1 mol��L��1 NH3��H2O
D��0.1 mol��L��1 NH4Cl��0.1 mol��L��1 HCl
1 Lij�����Һ�����ܺ��е��������±���
| ���ܴ������е������� | H����K����Mg2����Al3����NH4+��Fe2����Fe3�� |
| ���ܴ������е������� | Cl����Br����I����CO32����AlO2�� |
(1)������Һ����μ���NaOH��Һ���������������ʵ���(n)�����NaOH��Һ�����(V)�Ĺ�ϵ��ͼ��ʾ�������Һ��һ�������е�������_________________________��
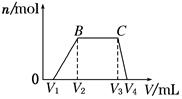
(2)BC�����ӷ���ʽΪ_______________________________________________��
(3)V1��V2��V3��V4֮��Ĺ�ϵΪ__________________________________________��
(4)����⣬����Һ�л����д�����Cl����Br����I��������1 L�û����Һ��ͨ��һ������Cl2����Һ��Cl����Br����I�������ʵ�����ͨ��Cl2�����(��״��)�Ĺ�ϵ�����ʾ��������ش��������⡣
| Cl2�����(��״��) | 2.8 L | 5.6 L | 11.2 L |
| n(Cl��) | 1.25 mol | 1.5 mol | 2 mol |
| n(Br��) | 1.5 mol | 1.4 mol | 0.9 mol |
| n(I��) | a mol | 0 | 0 |
�ٵ�ͨ��Cl2�����Ϊ2.8 Lʱ����Һ�з�����Ӧ�����ӷ���ʽΪ_________________��
��ԭ��Һ��Cl����Br����I�������ʵ���Ũ��֮��Ϊ_____________________________��
 2Fe(s)+3CO��g����H=+489.0KJ��mol
2Fe(s)+3CO��g����H=+489.0KJ��mol ��
��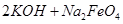 =
=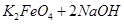 ��˵�����¶���
��˵�����¶���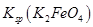 __________
__________ 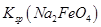 (�>����<��)��
(�>����<��)��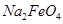 ��ȫת��Ϊ
��ȫת��Ϊ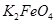 ���������Ƶôֲ�Ʒ
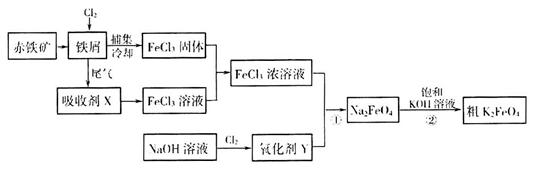
���������Ƶôֲ�Ʒ 3Zn��OH��2+2Fe��OH��3+4KOH���ŵ�ʱ��صĸ�����ӦʽΪ______________________________________��
3Zn��OH��2+2Fe��OH��3+4KOH���ŵ�ʱ��صĸ�����ӦʽΪ______________________________________��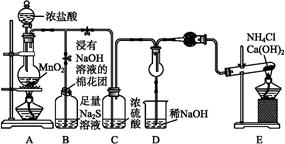
 2Cl2+2H2O,��ʵ���ȵ�ѭ�����á�
2Cl2+2H2O,��ʵ���ȵ�ѭ�����á�