��Ŀ����
8����1��FeCl3��H2S��Ӧ�����ӷ���ʽΪ2Fe3++H2S�T2Fe2++S��+2H+����2����������Һ�������ˮ��Ӧ�����ӷ���ʽΪAl3++3NH3��H2O�TAl��OH��3��+3NH4+��
��3��Ϊ̽��NH4HCO3��NaOH�ķ�Ӧ�����ʵ�鷽�����£���0.1mol NH4HCO3����Һ�м���0.1mol NaOH����Ӧ��ȫ�μ��Ȼ���ϡ��Һ�����г�������NH4HCO3��NaOH�ķ�Ӧ����ΪHCO3-+OH-=CO32-+H2O��д���ӷ���ʽ����
���� ��1�������ӽ��������������ʣ��ݴ�д�������ӷ���ʽ��
��2����������Һ�еι�����ˮ������������������
��3�����г�����˵����Һ�к���CO32-����HCO3-�� OH-��Ӧ����CO32-��
��� �⣺��1��Fe3+���������ԣ��ܹ����������ӣ���Ӧ�����ӷ���ʽΪ��2Fe3++H2S�T2Fe2++S��+2H+��
�ʴ�Ϊ��2Fe3++H2S�T2Fe2++S��+2H+��
��2����������Һ�백ˮ��Ӧ������������������������泥����������������������÷�Ӧ����ʽΪ��Al2��SO4��3+3NH3��H2O�T2Al��OH��3��+3��NH4��2SO4�������ӷ�ӦΪ��Al3++3NH3��H2O�TAl��OH��3��+3NH4+��
�ʴ�Ϊ��Al3++3NH3��H2O�TAl��OH��3��+3NH4+��
��3����0.1mol NH4HCO3����Һ�м���0.1mol NaOH����Ӧ��ȫ�μ��Ȼ���ϡ��Һ�����г������ɣ���˵��̼��������������������ӷ�Ӧ����̼������ӣ���Ӧ�����ӷ���ʽΪ��HCO3-+OH-=CO32-+H2O��
�ʴ�Ϊ��HCO3-+OH-=CO32-+H2O��
���� ���⿼�������ӷ���ʽ����д����Ŀ�Ѷ��еȣ�ע���������ӷ���ʽ����дԭ��2��ע�ⰱˮ���ܽ�������������3����Ҫ�жϷ�Ӧ��������̼������ӣ�笠�����û�в��뷴Ӧ��

 �����Ļ���������人������ϵ�д�
�����Ļ���������人������ϵ�д� ���������ּ���ÿһ��ȫ�º�����ҵ��ϵ�д�
���������ּ���ÿһ��ȫ�º�����ҵ��ϵ�д� ��ٽ������½������������ϵ�д�
��ٽ������½������������ϵ�д�| �������� | �� �� | |
| A | KI������Һ�е�����ˮ��������ͨ��SO2����ɫ��ȥ | SO2����Ư���� |
| B | ����SnCl2��Һʱ���Ƚ�SnCl2��������ϡ���ᣬ��������ˮϡ�ͣ�������Լ�ƿ�м������������� | ����Sn2+ˮ�⣬����ֹSn2+������ΪSn4+ |
| C | ij��Һ�м��������ữ���Ȼ�����Һ���а�ɫ�������� | ����˵������Һ��һ������SO42- |
| D | ��Ũ�Ⱦ�Ϊ0.1mol•L-1��MgCl2��CuCl2�����Һ����μ��백ˮ������������ɫ���� | Ksp[Cu��OH��2]��Ksp[Mg��OH��2] |
| A�� | A | B�� | B | C�� | C | D�� | D |
| ��� | �� | �� | �� | �� |
| װ�� | 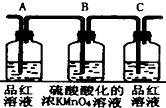 |  |  |  |
��2��ʵ��ʱ�ɹ۲쵽װ�â���Aƿ����Һ��ɫ��Cƿ����Һ����ɫ��Aƿ��Һ����������֤�����SO2��Bƿ��Һ�������ǽ�SO2ȫ���������գ�Cƿ��Һ��������ȷ��������SO2�ѱ�Bƿ��Һȫ��������
��3��װ�â������ӵĹ���ҩƷ����ˮ����ͭ����ȷ֤�IJ�����ˮ������ȷ��װ�â�������װ����λ�õ������Dz�������ͨ���١���ʱ�����ˮ���������Ԣڱ����ڢ١���֮�䣮
��4��װ�â�����ʢ��Һ�dz���ʯ��ˮ������֤�IJ�����CO2��
| A�� | ���ǵ��ʣ����ǵ��� | |
| B�� | ������ɷ�Ϊ����������ͷǽ��������� | |
| C�� | �Ȼ������ƵĻ���������Ȼ��� | |
| D�� | ���������Ǵ������������Ǽ� |
| A�� | Ũ���� | B�� | ϡ���� | C�� | Ũ���� | D�� | Ũ���� |
| A�� | ��������� | B�� | ���ȸ������ | C�� | ���ˮ | D�� | ������� |
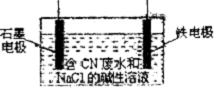 ������������ͭ���仯���������������й�ϵ���У�
������������ͭ���仯���������������й�ϵ���У�