��Ŀ����
9����5ƿ��ɫ�����Լ����ֱ���Ba��NO3��2��KCl��NaOH��CuSO4��Na2SO4����ֻ�ṩ����ˮ��ͨ�������ʵ�鲽��ɼ������ǣ�����д���пհף���1����ȡ���������Լ��ֱ����5֧�Թ��У�������������ˮ�����Թܣ��۲쵽����������Һ����ɫ������������ʵĻ�ѧʽ��CuSO4��
��2���ֱ�ȡδ�������Һ�������м��������Ѽ������Һ���۲쵽���������Ӧ�Ļ�ѧ����ʽ��
�������Թ����а�ɫ�������ɣ����ӷ���ʽ��Ba2++SO42-=BaSO4����
�������Թ�������ɫ�������ɣ���ѧ����ʽ��CuSO4+2NaOH�TCu��OH2����+2NaNO3�����������������֪��������������ʵĻ�ѧʽ��Ba��NO3��2��NaOH��
��3���ã�2������Ba��NO3��2���Լ���ʣ����������ʣ�
���� ��1��������ɫ���壬ֻ������ͭ����ˮ�õ���ɫ��Һ������Ϊ��ɫ��Һ��
��2������ͭ�����ᱵ��Ӧ���ɰ�ɫ���ᱵ����������ͭ��NaOH��Ӧ������ɫ������ͭ������
��3���������ᱵ�ɼ���KCl��Na2SO4���Դ������
��� �⣺��1��ͭ��������Һ������ɫ�����ȹ۲���Һ����ɫ������ɫ��������ͭ���ʴ�Ϊ����Һ����ɫ��CuSO4��
��2���ٽ�����ͭ����ʣ���������Һ�У����ְ�ɫ�����ɼ�������ᱵ����ѧ��Ӧ����ʽΪBa��NO3��2+CuSO4�TBaSO4��+Cu��NO3��2�����ӷ�ӦΪBa2++SO42-=BaSO4����
�ʴ�Ϊ��Ba2++SO42-=BaSO4����
�ڳ�����ɫ�����ɼ�����������ƣ���ѧ��Ӧ����ʽΪCuSO4+2NaOH�TCu��OH2����+2NaNO3�����������������֪��������������ʵĻ�ѧʽ��Ba��NO3��2��NaOH��
�ʴ�Ϊ��CuSO4+2NaOH�TCu��OH2����+2NaNO3��Ba��NO3��2��NaOH��
��3��Ȼ�����ᱵ����ʣ���������Һ�г��ְ�ɫ�������������ƣ�û����������Ȼ��أ��ʴ�Ϊ��Ba��NO3��2��
���� ���⿼�����ʵļ����飬Ϊ��Ƶ���㣬���շ����ķ�Ӧ�������۵Ĺ�ϵΪ���Ĺؼ������ط�����Ӧ�������Ŀ��飬��Ŀ�ѶȲ���

��1��ʵ������ȡ��Ȳ����ķ���ʽ��CaC2+2H2O��C2H2��+Ca��OH��2
��2���������������͵�����װʵ��װ�ã�
| ��� | 1 | 2 | 3 | 4 | 5 | 6 |
| ���ܼ����� |  |  | 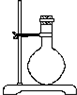 |  |  | 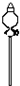 |
| ÿ����Ƥ���϶����������� | ||||||
��3���������Ӻú���ʵ��ʱ�������в�����ÿ�����ֻ����һ�Σ�
�ٳ�ȡһ������ʯ����������3�У�������Ƥ���� �ڼ��װ�õ������� ��������6��5��ע������ˮ�� �ܴ�����3�ָ�������ʱ����ȡ����4��ˮ�����������2��ˮ��������Բ��ƣ� ��������������6�Ļ�����ʹˮ��ε���������������ʱ���رջ�������ȷ����˳����B
A���٢ۢڢݢ�B���ڢ٢ۢݢ�C���٢ۢݢܢ�
��4����ʵ���в���������������ŵ���ζ���Ҳⶨ���ƫ��������Ϊ��ʯ�к����������壬����������ͭ��Һ������������Һ���Գ�ȥ��
| A�� | ���۳ɹ�����С����Һ���Ĵ��ζ� | |
| B�� | ��������� | |
| C�� | ��Һ�ײ�������ɫ������ | |
| D�� | ��Һ����� |
��1��ѡ����������ɱ�ʵ��������������У�������ƽ����ȷ��0.1g����ҩ�ס��ձ�������������ͷ�ιܡ�500 mL����ƿ�ȣ�
��2�����㣺��ҪNaOH���������Ϊ10.0 g��
��3���������ٳ���������NaOH����Ӧ����С�ձ��в�������ƽ�����̣�����̡������̡�����
��ijѧ������������ƽ����һ��С�ձ�������������ǰ��������ڱ�ߵ���̶ȴ�����ƽ��ֹʱ����ָ���ڷֶ��̵�ƫ��λ�ã�˵����ʱ�������С�ڣ�����ڡ���С�ڡ����ұߣ��ٶ����ճƵ�С�ձ�������Ϊ32.6 g���32.6g����32.61g���������á�������ʾ�������Ϸ������룬��������ʾ��������ȡ������ij������̣�������ͼ�б���ϻ��������λ�ã�����������ʾ����
| ��������/g | 50 | 20 | 20 | 10 | 5 |
| ������ȡ��������̣� |

��4���ܽ⡢��ȴ���ò�ʵ������Ҫʹ�ò������������ǽ�������ܽ⣮
��5��ת�ơ�ϴ�ӣ���ת��ʱӦʹ�ò�����������ϴ���ձ�2�Ρ�3����Ϊ�˱�֤����ȫ��ת��������ƿ�У�
��6�����ݡ�ҡ�ȣ����ݵIJ�����������ƿ�м�������ˮ����̶���1 cm��2 cmʱ�����ý�ͷ�ιܵμ�����ˮ��Һ��İ�Һ����ʹ��Ϳ̶������У�
��7��ijѧ��ʵ������NaOH��Һ��Ũ��Ϊ0.48mol•L-1��ԭ�������A��C������ĸ����
A��ʹ����ֽ�����������ƹ���
B������ƿ��ԭ��������������ˮ
C���ܽ����ձ�δ�����ϴ�ӣ�
| �ɷ� | ������g�� | Ħ��������g•mol-1�� |
| ���� | 50.00 | 342 |
| ����� | 0.50 | 174 |
| ��˾ƥ�� | 0.35 | 180 |
| ������� | 0.50 | 158 |
| ������ | 0.04 | 170 |
A������ B������� C��������� D��������
��2�����ʻ����ʼ�����K+����˾ƥ���в���K+�������ʵ���Ũ��Ϊ$\frac{0.50}{158}+\frac{2��0.50}{174}$ L•mol-1����ֻҪ��д���������ʽ������Ҫ��������
��3����������1L���ʻ����ʼ�������������У�1000mL����ƿ���ձ�����������ҩ�ס���ͷ�ιܡ�������ƽ�����ں�������д��ȱ���������ƣ�
��4������Һ���ƹ����У����в��������ƽ��û��Ӱ�����BD��
A����ҩƷ��������ƿ�м�����ˮ�ܽ�
B������ƿ��ʹ��ǰδ�����������������ˮ
C������ƿ��ʹ��ǰ�ո�������һ�����ʵ���Ũ�ȵ�NaCl��Һ��δϴ��
D������ҡ�Ⱥ���Һ���������ƿ�Ŀ̶��ߣ���δ���κδ�����
| A�� | 6.02��1023�� | B�� | 6.02�� | C�� | 98�� | D�� | 3.01��1023�� |
| A�� | ͬϵ��֮�以Ϊͬ���칹�� | |
| B�� | ����������������ֱ�ӽ�������л���״� | |
| C�� | ͬϵ��֮ǰ��Ԫ�ص���������һ����ͬ | |
| D�� | ����̼ͬԭ����������һ���ǻ�Ϊͬϵ��Ĺ�ϵ |