��Ŀ����
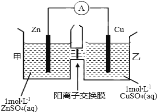
����Ŀ����Ǧ�������Ӷ��Ե缫���һ������CuSO4��Һ����ش��������⣺
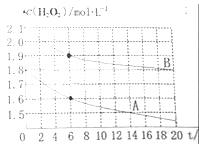
��1��Ǧ���طŵ�ʱ�����ĵ缫��ӦʽΪ________________________________��
��2�����Ե缫���CuSO4��Һ�����ӷ���Ϊ_____________________��
��3�������һ��ʱ����������Һ�м���1molCu��OH��2��CuCO3������ʹ����Һ�ָ���ԭ����Ũ�Ⱥ���������ڸõ�������ת�Ƶ��ӵ���ĿΪ___________���õ�������Ǧ���������ĵ�H2SO4�͵��������ɵ�H2SO4�����ʵ���֮��Ϊ____________��
���𰸡���1��PbO2+2e-+4H++SO42-=PbSO4+2H2O ��2��2Cu2++2H2O![]() 2Cu+O2��+4H+ ��3��6NA 2:1
2Cu+O2��+4H+ ��3��6NA 2:1
��������
�����������1��Ǧ���طŵ�ʱ��������ΪPbO2�������ĵ缫��ӦʽΪPbO2+2e-+4H++SO42-=PbSO4+2H2O��
��2�����ö��Ե缫���CuSO4��Һʱ������Cu�������������������ų���������Ӧ�����ӷ���Ϊ2Cu2++2H2O![]() 2Cu+O2��+4H+��
2Cu+O2��+4H+��
��3��Cu��OH��2��CuCO3��ϡ���ᷴӦ��������ͭ��ˮ�Ͷ�����̼�����Լ���1molCu��OH��2��CuCO3�൱�ڼ���2molCuO��1molH2O������������֪��������ͭ���Ӻ������ӷŵ硢���������������ӷŵ磬����Cuԭ�ӡ�Hԭ���غ������������n��Cu��=2mol��n��H2��=1mol����ת�Ƶ��ӵ����ʵ���=2mol��2+1mol��2=6mol������������ת�Ƶ��ӵ���ĿΪ6NA��1molCu��OH��2��CuCO3�ܺ�2molH2SO4��ȫ��Ӧ����֪�������ɵ�H2SO4Ϊ2mol������PbO2+Pb+2H2SO4=2PbSO4+2H2O��֪����������ת��6mol��������6molH2SO4�����������Ǧ���������ĵ�H2SO4�͵��������ɵ�H2SO4�����ʵ���֮��Ϊ6mol��3mol=2:1��