��Ŀ����
����Ŀ��![]() ��
��![]() ��
��![]() ��
��![]() ����Ԫ��ԭ�ӵĺ˵���������������ǵļ����ӵĵ��Ӳ�����ͬ��������������Ϊ8��
����Ԫ��ԭ�ӵĺ˵���������������ǵļ����ӵĵ��Ӳ�����ͬ��������������Ϊ8��![]() Ԫ��ԭ�ӵ�
Ԫ��ԭ�ӵ�![]() ���������
���������![]() ��
��![]() �������֮����ȣ�
�������֮����ȣ�![]() Ԫ��ԭ�ӵ�
Ԫ��ԭ�ӵ�![]() ��
��![]() �������֮�͵��ڵ���������һ�롣�Իش��������⡣
�������֮�͵��ڵ���������һ�롣�Իش��������⡣
��1������Ԫ�صķ���������![]() ______��
______��![]() ______��
______��![]() ______��
______��![]() ______�����ǵ�ԭ�Ӱ뾶�ɴ�С��˳����______����Ԫ�ط��ű�ʾ����
______�����ǵ�ԭ�Ӱ뾶�ɴ�С��˳����______����Ԫ�ط��ű�ʾ����
��2����д������Ԫ�ص����ӽṹʾ��ͼ��![]() ______��
______��![]() ______��
______��![]() ______��
______��![]() ______�����ǵ����Ӱ뾶�ɴ�С��˳����______�������ӷ��ű�ʾ����
______�����ǵ����Ӱ뾶�ɴ�С��˳����______�������ӷ��ű�ʾ����
��3���ֱ�д������Ԫ�ص�����������Ӧˮ����Ļ�ѧʽ��______��
��4���ֱ�д��![]() ��
��![]() ��Ԫ�ص���̬�⻯��Ļ�ѧʽ��______��
��Ԫ�ص���̬�⻯��Ļ�ѧʽ��______��
���𰸡�![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]() ��
��![]() ��
��![]() ��
��![]()
![]() ��
��![]()
��������
����ԭ�ӽṹ����Ԫ�ص����࣬����Ԫ�������ɷ������
AԪ��ԭ�ӵĵڶ�����������һ�������������֮����ȣ��ڶ��������Ϊ8����һ�������Ϊ2����Aԭ�ӵ�����������Ϊ6��AΪ��Ԫ�أ�DԪ��ԭ�ӵĵ�һ���ڶ��������֮��Ϊ10�����ڵ���������һ�룬��Dԭ�ӵĵ�������Ϊ20��DΪ��Ԫ�أ�A��B��C��D����Ԫ��ԭ�ӵĺ˵�����������ӣ����ǵļ����ӵĵ��Ӳ�����ͬ��������������Ϊ8����A��B��C��D�ֱ�ΪS��Cl��K��Ca��
��1���������Ϸ�����֪������Ԫ�صķ���������A��S��B��Cl��C��K��D��Ca��ԭ�Ӻ�����Ӳ���Խ�࣬ԭ�Ӱ뾶Խ��ͬһ���ڴ�����ԭ�Ӱ뾶���μ�С��������ǵ�ԭ�Ӱ뾶�ɴ�С��˳���ǣ�![]() ���ʴ�Ϊ��
���ʴ�Ϊ��![]() ��
��![]() ��
��![]() ��
��![]() ��
��![]() ��
��
��2�������Ӻ���������Ϊ16��������18�����ӣ����ӽṹʾ��ͼΪ��![]() �������Ӻ���������Ϊ17��������18�����ӣ����ӽṹʾ��ͼΪ��
�������Ӻ���������Ϊ17��������18�����ӣ����ӽṹʾ��ͼΪ��![]() �������Ӻ���������Ϊ19��������18�����ӣ����ӽṹʾ��ͼΪ��
�������Ӻ���������Ϊ19��������18�����ӣ����ӽṹʾ��ͼΪ��![]() �������Ӻ���������Ϊ20��������18�����ӣ����ӽṹʾ��ͼΪ��
�������Ӻ���������Ϊ20��������18�����ӣ����ӽṹʾ��ͼΪ��![]() �����ӵĺ�������Ų���ͬʱ��ԭ������Խ�����Ӱ뾶ԽС��������Ӱ뾶�ɴ�С��˳���ǣ�
�����ӵĺ�������Ų���ͬʱ��ԭ������Խ�����Ӱ뾶ԽС��������Ӱ뾶�ɴ�С��˳���ǣ�![]() ���ʴ�Ϊ��
���ʴ�Ϊ��![]() ��
��![]() ��
��![]() ��
�� ![]() ��
��![]() ��
��
��3��S��Cl��K��Ca������ϼ۷ֱ�Ϊ+6��+7��+1��+2������������Ӧˮ����Ļ�ѧʽ�ֱ�Ϊ��![]() ��
��![]() ��
��![]() ��
��![]() ���ʴ�Ϊ��
���ʴ�Ϊ��![]() ��
��![]() ��
��![]() ��
��![]() ��
��
��4��A��B��Ԫ�ص���̬�⻯��Ļ�ѧʽ�ֱ�Ϊ![]() ��
��![]() ���ʴ�Ϊ��
���ʴ�Ϊ��![]() ��
��![]() ��
��

����Ŀ��ij�����̿����Ҫ�ɷ�ΪMnCO3��������������FeCO3��CaCO3��Al2O3�����ʡ���ҵ�������̿�Ϊԭ���Ʊ��ߴ���̼���̵�������ͼ��ʾ��
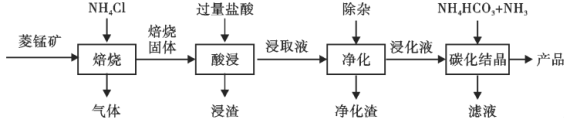
��֪��MnCO3+2NH4Cl=MnCl2+CO2��+2NH3��+H2O��
��ؽ�������[c0��Mn+��=0.1mol��L��1]�γ��������������pH��Χ���£�
�������� | Mn2+ | Fe2+ | Fe3+ | Al3+ |
��ʼ������pH | 8.1 | 6.3 | 1.5 | 3.4 |
������ȫ��pH | 10.1 | 8.3 | 2.8 | 4.7 |
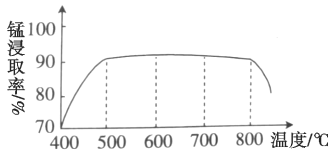
��1������ʱ�¶ȶ��̽�ȡ�ʵ�Ӱ����ͼ������ʱ���˵��¶�Ϊ___________���ң�800�������̵Ľ�ȡ��ƫ�ͣ����ܵ�ԭ����___________��
��2�����������������ټ�������MnO2����������MnO2��������____________��������Ӧ�����ӷ���ʽΪ___________��
�ڼӰ�ˮ��pH����Һ��pH��ΧӦ����Ϊ___________��8.1֮�䡣���ɵij�����Ҫ��___________��
�ۼ���MnF2��������ȥCa2+������Һ��ȹ��ߣ�Ca2+��������ȫ��ԭ����___________��
��3��̼���ᾧ������MnCO3�����ӷ���ʽΪ____________��
����Ŀ�������(ZrO2)���Ͼ��и�Ӳ�ȡ����۵㡢��ǿ�ȡ������ԡ����ߵ���ĥ�Լ��ͻ�ѧ��ʴ�Ե��������ﻯ���ܡ����Ӣʯ(��Ҫ�ɷ�Ϊ ZrSiO4����������Al2O3��SiO2��Fe2O3������)Ϊԭ��ͨ�����۷��Ʊ������(ZrO2)���������£�
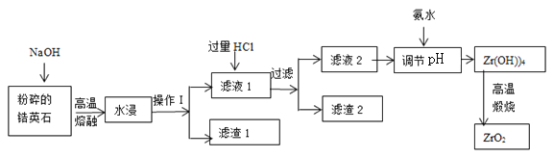
25��ʱ���й�������ˮ��Һ�г���ʱ��pH���ݣ�
Fe(OH)3 | Zr(OH)4 | Al(OH)3 | |
��ʼ����ʱpH | 1.9 | 2.2 | 3.4 |
������ȫʱpH | 3.2 | 3.2 | 4.7 |
��ش��������⣺
(1)������ּ�����ѧ��Ӧ���ʵĴ�ʩ��________________________________��
(2)����I��������__________________������1�ɷ�Ϊ_________������2�ijɷ�Ϊ_____________��
(3)�Ӣʯ��������������ת��ΪNa2ZrO3��д���÷�Ӧ�Ļ�ѧ����ʽ��____________________��
(4)������pH��ʱ�����ʵ�pH��Χ��__________________��Ϊ�˵õ�����ZrO2��Zr(OH)4��Ҫϴ�ӣ�����Zr(OH)4�Ƿ�ϴ�Ӹɾ��ķ�����__________________��
(5)д�����������������̵Ļ�ѧ����ʽ________________________________������ZrO2�����ʣ��Ʋ���һ����;________________________________��