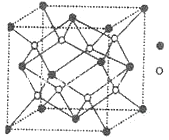
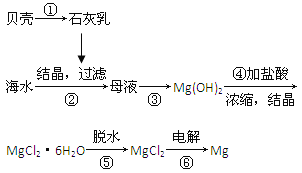
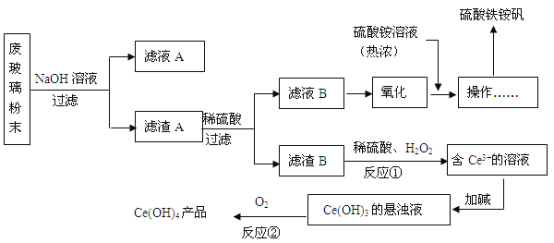
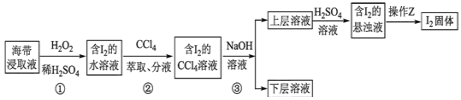
��Ŀ����
����Ŀ����������(Na2FeO4)���к�ǿ��������,��һ�����͵���ɫ��ˮ����������ҵ����������(��Ҫ�ɷ���FeCO3������SiO2)Ϊԭ���Ʊ��������������������£�
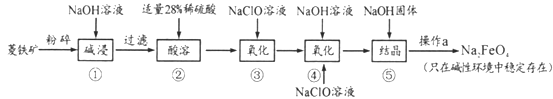
(1)Na2FeO4����Ԫ�صĻ��ϼ�Ϊ___,������������ɱ������ʱ�Ļ�ѧ��Ӧ����Ϊ____(����������ԭ��Ӧ���������ֽⷴӦ���������Ϸ�Ӧ��)��
(2)������������,������м��ʱ�ܷ��ýϱ��˵�Ca(OH)2���NaOH_____(������������������),������___________��
(3)�������28%��ϡ������Ҫ��98%��Ũ��������,����ʱ���貣����������Ͳ��,��____(����ĸ���)��������м���Fe2+ȫ��ת����Fe3+�ķ����ǣ�__________��
A������ƿ B���ձ� C����ƿ D�������� E����ʽ�ζ���;
(4)������г�����Na2FeO4��,����NaCl����,�����ӷ���ʽΪ_________����֪���������Na2FeO4��Һ�м��������������ƹ���õ�����Һ,�����a������Ϊ_____��
(5)������,ÿ���0.5mol��FeO42-���� NaClO��������Ϊ_____��
���𰸡�+6 ������ԭ��Ӧ ���� CaSiO3 ������ˮ���������SiO2������������Ҳ�ɣ� BD ȡ����������Һ���Թܣ���������K3Fe(CN)6��Һ��������ɫ��������Fe2�� �Ѿ�ȫ��ת����Fe3�� 2Fe3����3ClO����10OH��=2FeO42����3Cl����5H2O ���� 74.5g
��������
��ҵ����������Ҫ�ɷ���FeCO3������SiO2������NaOH�ܽ�SiO2��ȥ�����������28%��ϡ�����ܽ������������ӣ���NaClO���������������������ӣ���Ȼ�����NaOH��NaClO������Ӧ��2Fe3��+3ClO��+10OH��=2FeO42��+3Cl��+5H2O���õ��������ƣ�Na2FeO4����Һ�������ṹ����Ũ������ȴ�ᾧ�õ���Ʒ�������ơ�
��1��Na2FeO4�л��ϼ۴�����Ϊ�㣻
��2��CaSiO3 ������ˮ��
��3����������һ����������Һ�IJ��裺���㡢�������ܽ⣬ȷ�����õ�������
����K3Fe(CN)6��Һ������Fe2+��
��4��������г�����Na2FeO4�⣬����NaCl���ɣ����������غ�д�����ӷ���ʽ��
���������Һ��Ӧ�ù��ˣ�
��5�������ϣ��ɵ����غ㣬����+2�۱��+6�ۣ�����ϵʽFeO42-��2NaClO���㡣
��1��Na2FeO4�л��ϼ۴�����Ϊ�㣬��FeԪ�صĻ��ϼ�Ϊ+6�ۣ�
�������ƾ��к�ǿ�������ԣ�������������ɱ������ʱ�Ļ�ѧ��Ӧ����Ϊ������ԭ��Ӧ��
��2����NaOH�ܽ�SiO2���ɹ��������ܽ⣬�Ӷ���FeCO3�������ȥ�������ýϱ��˵�Ca(OH)2���NaOH��CaSiO3 ������ˮ���������SiO2��
��3����������һ����������Һ�IJ��裺���㡢�������ܽ⣬ȷ�����õ���������ͷ�ιܡ���Ͳ�����������ձ�����ѡ��BD��
������м���Fe2+ȫ��ת����Fe3+�ķ����ǣ�ȡ����������Һ���Թܣ���������K3Fe(CN)6��Һ��������ɫ��������Fe2�� �Ѿ�ȫ��ת����Fe3����
��4��������г�����Na2FeO4�⣬����NaCl����,�����ӷ���ʽΪ2Fe3����3ClO����10OH��=2FeO42����3Cl����5H2O��
��֪���������Na2FeO4��Һ�м��������������ƹ���õ�����Һ�����������Һ��Ӧ�ù��ˣ������a������Ϊ���ˡ�
��5�������ϣ��ɵ����غ㣬����+2�۱��+6�ۣ�����ϵʽFeO42-��2NaClO���㣬ÿ���0.5mol��FeO42-���� NaClO��������Ϊ74.5g��

 Сѧѧϰ�ð���ϵ�д�
Сѧѧϰ�ð���ϵ�д� Сѧͬ�����������ܾ�ϵ�д�
Сѧͬ�����������ܾ�ϵ�д�����Ŀ������ʵ�������ȷ���ǣ� ��
���� | ���� | ���� | |
A | ��SO2ͨ����ˮ | ��ˮ��ɫ | SO2����Ư���� |
B | ����ϩͨ������KMnO4��Һ | ����KMnO4��Һ��ɫ | ��ϩ���л�ԭ�� |
C | ������ʯ��ˮ����ij��Һ | ���ְ�ɫ���� | ����Һ�д���CO32- |
D | ����������Һ����ij��Һ | ���ְ�ɫ���� | ����Һ�д���Cl- |
A.AB.BC.CD.D
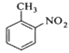
����Ŀ���������ױ���ҽҩ��Ⱦ�ϵȹ�ҵ��һ����Ҫ�л��м��壬������Ũ����Ϊ��������Ũ����Ϊ������ͨ���ױ���������Ӧ�Ʊ���
![]()
![]()
![]() +
+ +
+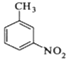
һ���µ��Ʊ��������ױ���ʵ�鷽���ǣ��Է�������Ϊ������������NaHSO4Ϊ����(��ѭ��ʹ��)����CCl4��Һ�У�����������(����ˮ����)��45�淴Ӧ1h ����Ӧ�������ˣ���Һ�ֱ���5% NaHCO3��Һ��ˮϴ�����ԣ��پ������ᴿ�õ��������ױ���
(1)����ʵ���й��˵�Ŀ����___________��
(2)��Һ�ڷ�Һ©����ϴ�Ӿ��ú��л��㴦��________��(����������'����)��
(3)5% NaHCO3��Һϴ�ӵ�Ŀ����__________
(4)���и����˴������༰�����Լױ�������ӦӰ���ʵ������
���� | n(����)/n(�ױ�) | ���������и����칹����������(%) | �ܲ���(%) | ||
�������ױ� | �������ױ� | �������ױ� | |||
ŨH2SO4 | 1.0 | 35.6 | 60.2 | 4.2 | 98.0 |
1.2 | 36.5 | 59.5 | 4.0 | 99.8 | |
NaHSO4 | 0.15 | 44.6 | 55.1 | 0.3 | 98.9 |
0.25 | 46.3 | 52.8 | 0.9 | 99.9 | |
0.32 | 47.9 | 51.8 | 0.3 | 99.9 | |
0.36 | 45.2 | 54.2 | 0.6 | 99.9 | |
��NaHSO4���Ʊ��������ױ�ʱ��������ױ���������ʵ���֮��Ϊ_______________��
���ɼױ������õ��ĸ��ֲ���ĺ�����֪���ױ�������Ӧ���ص���_________________��
����Ũ������ױ�������ȣ�NaHSO4���ױ��������ŵ���_____________��