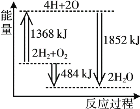
ΧβΡΩΡΎ»ί
ΓΨΧβΡΩΓΩ≤ίΥα―«ΧζΨßΧε![]() «“Μ÷÷ΜΤ…ΪΡ―»ή”ΎΥ°ΒΡΙΧΧεΘ§ ή»»“ΉΖ÷ΫβΘ§ «…ζ≤ζο°Βγ≥ΊΓΔΆΩΝœΓΔΉ≈…ΪΦΝ“‘ΦΑΗ–Ιβ≤ΡΝœΒΡ‘≠≤ΡΝœΓΘ
«“Μ÷÷ΜΤ…ΪΡ―»ή”ΎΥ°ΒΡΙΧΧεΘ§ ή»»“ΉΖ÷ΫβΘ§ «…ζ≤ζο°Βγ≥ΊΓΔΆΩΝœΓΔΉ≈…ΪΦΝ“‘ΦΑΗ–Ιβ≤ΡΝœΒΡ‘≠≤ΡΝœΓΘ
IΓΔ÷Τ±Η≤ίΥα―«ΧζΨßΧεΘΚ
![]() ≥Τ»ΓΝρΥα―«ΧζοßΨßΧε
≥Τ»ΓΝρΥα―«ΧζοßΨßΧε![]() Θ§Ζ≈»κ
Θ§Ζ≈»κ![]() »ΐΨ±…’ΤΩ÷–Θ§Φ”»κ10ΒΈΉσ”“
»ΐΨ±…’ΤΩ÷–Θ§Φ”»κ10ΒΈΉσ”“![]() »ή“ΚΚΆ
»ή“ΚΚΆ![]() ’τΝσΥ°Φ”»»»ήΫβΘΜ
’τΝσΥ°Φ”»»»ήΫβΘΜ
![]() Φ”»κ
Φ”»κ![]() ±ΞΚΆ≤ίΥα»ή“ΚΘ§Φ”»»ΫΝΑη÷ΝΖ–Θ§ΆΘ÷ΙΦ”»»Θ§Ψ≤÷ΟΘΜ
±ΞΚΆ≤ίΥα»ή“ΚΘ§Φ”»»ΫΝΑη÷ΝΖ–Θ§ΆΘ÷ΙΦ”»»Θ§Ψ≤÷ΟΘΜ
![]() ¥ΐΜΤ…ΪΨßΧε
¥ΐΜΤ…ΪΨßΧε![]() ≥ΝΒμΚσ«ψΈωΘ§œ¥Β”Θ§Φ”»κ‘Φ
≥ΝΒμΚσ«ψΈωΘ§œ¥Β”Θ§Φ”»κ‘Φ![]() ’τΝσΥ°ΫΝΑη≤ΔΈ¬»»
’τΝσΥ°ΫΝΑη≤ΔΈ¬»»![]() Θ§Ψ≤÷ΟΘ§Τζ»Ξ…œ≤ψ«ε“ΚΘ§Φ¥ΒΟΜΤ…Ϊ≤ίΥα―«ΧζΨßΧεΓΘ
Θ§Ψ≤÷ΟΘ§Τζ»Ξ…œ≤ψ«ε“ΚΘ§Φ¥ΒΟΜΤ…Ϊ≤ίΥα―«ΧζΨßΧεΓΘ

ΜΊ¥πœ¬Ν–Έ ΧβΘΚ
Θ®1Θ©“«ΤςCΒΡΟϊ≥Τ «_____________ΘΜ“«ΤςBΒΡΉς”Ο «_____________
Θ®2Θ©ΝρΥα―«ΧζοßΨßΧε”ΟΝρΥα»ήΫβΒΡ‘≠“ρ « _____________________
IIΓΔΧΫΨΩ≤ίΥα―«ΧζΒΡ¥ΩΕ»ΘΚ
![]() ΉΦ»Ζ≥Τ»Γ
ΉΦ»Ζ≥Τ»Γ![]() ≤ίΥα―«ΧζΙΧΧε―υΤΖ
≤ίΥα―«ΧζΙΧΧε―υΤΖ![]() Κ§”–≤ίΥαοß‘”÷
Κ§”–≤ίΥαοß‘”÷ ![]() Θ§»ή”Ύ
Θ§»ή”Ύ![]() »ή“Κ÷–Θ§‘Ύ
»ή“Κ÷–Θ§‘Ύ![]() Υ°‘ΓΦ”»»Θ§”Ο
Υ°‘ΓΦ”»»Θ§”Ο![]() »ίΝΩΤΩ≈δ≥…
»ίΝΩΤΩ≈δ≥…![]() »ή“ΚΘΜ
»ή“ΚΘΜ
![]() »Γ…œ ω»ή“Κ
»Γ…œ ω»ή“Κ![]() Θ§”Ο
Θ§”Ο![]() ±ξΉΦ»ή“Κ
±ξΉΦ»ή“Κ![]() ≈®Ε»ΈΣ
≈®Ε»ΈΣ![]() ΒΈΕ®»ΐ¥ΈΘ§ΤΫΨυœϊΚΡ
ΒΈΕ®»ΐ¥ΈΘ§ΤΫΨυœϊΚΡ![]() ΘΜ
ΘΜ
![]() Φ”»κ ΝΩ–ΩΖέΚΆ
Φ”»κ ΝΩ–ΩΖέΚΆ![]() »ή“ΚΘ§Ζ¥”Π“ΜΕΈ ±ΦδΚσΘ§»Γ1ΒΈ ‘“ΚΖ≈‘ΎΒψΒΈΑε…œΦλ―ιΘ§÷Ν
»ή“ΚΘ§Ζ¥”Π“ΜΕΈ ±ΦδΚσΘ§»Γ1ΒΈ ‘“ΚΖ≈‘ΎΒψΒΈΑε…œΦλ―ιΘ§÷Ν![]() ΦΪΈΔΝΩΘΜ
ΦΪΈΔΝΩΘΜ
![]() Ιΐ¬Υ≥ΐ»Ξ–ΩΖέΘ§¬Υ“Κ ’Φ·‘ΎΝμ“ΜΗωΉΕ–ΈΤΩ÷–Θ§ΫΪ¬Υ÷ΫΦΑ≤–”ύΈο≥δΖ÷œ¥Β”Θ§œ¥Β”“Κ≤Δ»κ¬Υ“Κ÷–Θ§‘Ό≤Ι≥δ‘Φ
Ιΐ¬Υ≥ΐ»Ξ–ΩΖέΘ§¬Υ“Κ ’Φ·‘ΎΝμ“ΜΗωΉΕ–ΈΤΩ÷–Θ§ΫΪ¬Υ÷ΫΦΑ≤–”ύΈο≥δΖ÷œ¥Β”Θ§œ¥Β”“Κ≤Δ»κ¬Υ“Κ÷–Θ§‘Ό≤Ι≥δ‘Φ![]() »ή“ΚΘ§ΦΧ–χ”Ο
»ή“ΚΘ§ΦΧ–χ”Ο![]() ±ξΉΦ»ή“Κ
±ξΉΦ»ή“Κ![]() ≈®Ε»ΈΣ
≈®Ε»ΈΣ![]() ΒΈΕ®÷Ν÷’ΒψΘ§Ι≤ΤΫ––≤βΕ®»ΐ¥ΈΘ§ΤΫΨυœϊΚΡ
ΒΈΕ®÷Ν÷’ΒψΘ§Ι≤ΤΫ––≤βΕ®»ΐ¥ΈΘ§ΤΫΨυœϊΚΡ![]() ΓΘ
ΓΘ
ΜΊ¥πœ¬Ν–Έ ΧβΘΚ
Θ®3Θ©‘Ύ![]() ÷–»ή“Κ÷–ΖΔ…ζ
÷–»ή“Κ÷–ΖΔ…ζ![]() Θ§ΜΙ”–Νμ“Μ―θΜ·ΜΙ‘≠Ζ¥”ΠΘ§–¥≥ωΤδάκΉ”ΖΫ≥Χ Ϋ_______________________Θ§”Ο
Θ§ΜΙ”–Νμ“Μ―θΜ·ΜΙ‘≠Ζ¥”ΠΘ§–¥≥ωΤδάκΉ”ΖΫ≥Χ Ϋ_______________________Θ§”Ο![]() ±ξΉΦ»ή“ΚΒΈΕ®÷Ν÷’ΒψΒΡœ÷œσ «__________________ΓΘ
±ξΉΦ»ή“ΚΒΈΕ®÷Ν÷’ΒψΒΡœ÷œσ «__________________ΓΘ
Θ®4Θ©Φ”»κ ΝΩ–ΩΖέΒΡΡΩΒΡ «______________________________________ΘΜ≤Ϋ÷η![]() ÷–Φλ―ιΤδ÷–ΈΔΝΩΒΡ
÷–Φλ―ιΤδ÷–ΈΔΝΩΒΡ![]() ΒΡ ‘ΦΝΟϊ≥Τ «______________»ή“Κ
ΒΡ ‘ΦΝΟϊ≥Τ «______________»ή“Κ
Θ®5Θ©ΦΤΥψ![]() ΙΧΧε―υΤΖΒΡ¥ΩΕ»ΈΣ______________
ΙΧΧε―υΤΖΒΡ¥ΩΕ»ΈΣ______________![]() ”ΟΚ§cΓΔ
”ΟΚ§cΓΔ![]() ΓΔ
ΓΔ![]() ΒΡ ΫΉ”±μ ΨΘ§≤Μ±ΊΜ·Φρ
ΒΡ ΫΉ”±μ ΨΘ§≤Μ±ΊΜ·Φρ![]()
ΓΨ¥πΑΗΓΩΚψ―ΙΒΈ“Κ¬©ΕΖ άδΡΐΜΊΝς “÷÷Τ![]() ΒΡΥ°Ϋβ
ΒΡΥ°Ϋβ ![]() ΉνΚσ“ΜΒΈ±ξΉΦ»ή“ΚΦ”»κΚσΘ§ΉΕ–ΈΤΩ÷–ΒΡ»ή“Κ―’…Ϊ”…ΜΤ…Ϊ±δΈΣΒ≠ΉœΚλ…ΪΘ§«“ΑκΖ÷÷”ΡΎ≤ΜΆ …Ϊ ΫΪ
ΉνΚσ“ΜΒΈ±ξΉΦ»ή“ΚΦ”»κΚσΘ§ΉΕ–ΈΤΩ÷–ΒΡ»ή“Κ―’…Ϊ”…ΜΤ…Ϊ±δΈΣΒ≠ΉœΚλ…ΪΘ§«“ΑκΖ÷÷”ΡΎ≤ΜΆ …Ϊ ΫΪ![]() ΜΙ‘≠ΈΣ
ΜΙ‘≠ΈΣ![]() Νρ«ηΜ·ΦΊ
Νρ«ηΜ·ΦΊ ![]() ΓΘ
ΓΘ
ΓΨΫβΈωΓΩ
Θ®1Θ©ΗυΨίΉΑ÷ΟΆΦΜΊ¥π“«ΤςCΒΡΟϊ≥ΤΘΜ“«ΤςB «άδΡΐΙήΘΜ
Θ®2Θ©ΗυΨί![]() “ΉΥ°ΫβΖ÷Έω‘≠“ρΘΜ
“ΉΥ°ΫβΖ÷Έω‘≠“ρΘΜ
Θ®3Θ©≤ίΥαΡή±ΜΥα–‘ΗΏΟΧΥαΦΊ»ή“Κ―θΜ·ΘΜ―«ΧζάκΉ”ΓΔ¥ΉΥαΗυάκΉ”Άξ»ΪΖ¥”ΠΚσΘ§ΒΈ»κΒΡΗΏΟΧΥαΦΊ Θ”ύΓΘ
Θ®4Θ©–ΩΖέΩ…“‘ΫΪ![]() ΜΙ‘≠ΈΣ
ΜΙ‘≠ΈΣ![]() ΘΜ
ΘΜ![]() ”ωΝρ«ηΜ·ΦΊ»ή“Κœ‘―ΣΚλ…ΪΘΜ
”ωΝρ«ηΜ·ΦΊ»ή“Κœ‘―ΣΚλ…ΪΘΜ
Θ®5Θ©ΗυΨί![]() ΦΤΥψ―υΤΖ¥ΩΕ»ΓΘ
ΦΤΥψ―υΤΖ¥ΩΕ»ΓΘ
![]() ΗυΨίΉΑ÷ΟΆΦΩ…÷ΣΘ§“«ΤςCΒΡΟϊ≥Τ «Κψ―ΙΒΈ“Κ¬©ΕΖΘΜ“«ΤςB «άδΡΐΙήΘ§ΤδΉς”Ο «άδΡΐΜΊΝςΘΜ
ΗυΨίΉΑ÷ΟΆΦΩ…÷ΣΘ§“«ΤςCΒΡΟϊ≥Τ «Κψ―ΙΒΈ“Κ¬©ΕΖΘΜ“«ΤςB «άδΡΐΙήΘ§ΤδΉς”Ο «άδΡΐΜΊΝςΘΜ
![]() ΝρΥαΩ…“‘“÷÷Τ
ΝρΥαΩ…“‘“÷÷Τ![]() ΒΡΥ°ΫβΘ§Υυ“‘ΝρΥα―«ΧζοßΨßΧε”ΟΝρΥα»ήΫβΘΜ
ΒΡΥ°ΫβΘ§Υυ“‘ΝρΥα―«ΧζοßΨßΧε”ΟΝρΥα»ήΫβΘΜ
![]() ‘Ύ
‘Ύ![]() ÷–»ή“Κ÷–ΜΙΚ§”–≤ίΥαΘ§≤ίΥα±ΜΗΏΟΧΥαΦΊ―θΜ·Θ§ΖΔ…ζΖ¥”ΠΒΡάκΉ”ΖΫ≥Χ ΫΈΣ
÷–»ή“Κ÷–ΜΙΚ§”–≤ίΥαΘ§≤ίΥα±ΜΗΏΟΧΥαΦΊ―θΜ·Θ§ΖΔ…ζΖ¥”ΠΒΡάκΉ”ΖΫ≥Χ ΫΈΣ![]() Θ§”Ο
Θ§”Ο![]() ±ξΉΦ»ή“ΚΒΈΕ®÷Ν÷’ΒψΒΡœ÷œσ «ΉνΚσ“ΜΒΈ±ξΉΦ»ή“ΚΦ”»κΚσΘ§ΉΕ–ΈΤΩ÷–ΒΡ»ή“Κ―’…Ϊ”…ΜΤ…Ϊ±δΈΣΒ≠ΉœΚλ…ΪΘ§«“ΑκΖ÷÷”ΡΎ≤ΜΆ …ΪΘΜ
±ξΉΦ»ή“ΚΒΈΕ®÷Ν÷’ΒψΒΡœ÷œσ «ΉνΚσ“ΜΒΈ±ξΉΦ»ή“ΚΦ”»κΚσΘ§ΉΕ–ΈΤΩ÷–ΒΡ»ή“Κ―’…Ϊ”…ΜΤ…Ϊ±δΈΣΒ≠ΉœΚλ…ΪΘ§«“ΑκΖ÷÷”ΡΎ≤ΜΆ …ΪΘΜ
![]() Φ”»κ ΝΩ–ΩΖέΒΡΡΩΒΡ «ΫΪ
Φ”»κ ΝΩ–ΩΖέΒΡΡΩΒΡ «ΫΪ![]() ΜΙ‘≠ΈΣ
ΜΙ‘≠ΈΣ![]() ΘΜ≤Ϋ÷η
ΘΜ≤Ϋ÷η![]() ÷–Φλ―ιΤδ÷–ΈΔΝΩΒΡ
÷–Φλ―ιΤδ÷–ΈΔΝΩΒΡ![]() ΒΡ ‘ΦΝΟϊ≥Τ «Νρ«ηΜ·ΦΊ»ή“ΚΘ§»τΒΈ»κΝρ«ηΜ·ΦΊ»ή“Κ ±»ή“Κ±δΈΣ―ΣΚλ…Ϊ‘ρΚ§”–
ΒΡ ‘ΦΝΟϊ≥Τ «Νρ«ηΜ·ΦΊ»ή“ΚΘ§»τΒΈ»κΝρ«ηΜ·ΦΊ»ή“Κ ±»ή“Κ±δΈΣ―ΣΚλ…Ϊ‘ρΚ§”–![]() ΘΜ
ΘΜ
![]() ΗυΨίΖ¥”Π
ΗυΨίΖ¥”Π![]() Ω…÷ΣΘ§
Ω…÷ΣΘ§![]() ΙΧΧε―υΤΖΒΡ¥ΩΕ»ΈΣ
ΙΧΧε―υΤΖΒΡ¥ΩΕ»ΈΣ ΓΘ
ΓΘ

 ’ψΫ≠÷°–«ΩΈ ±”≈Μ·Ής“ΒœΒΝ–¥πΑΗ
’ψΫ≠÷°–«ΩΈ ±”≈Μ·Ής“ΒœΒΝ–¥πΑΗ ΦΛΜνΥΦΈ§”≈Φ”ΩΈΧΟœΒΝ–¥πΑΗ
ΦΛΜνΥΦΈ§”≈Φ”ΩΈΧΟœΒΝ–¥πΑΗΓΨΧβΡΩΓΩ25Γφ ±Θ§»ΐ÷÷ΥαΒΡΒγάκΤΫΚβ≥Θ ΐ»γœ¬ΘΚ
Μ·―ß Ϋ | CH3COOH | H2CO3 | HClO |
ΒγάκΤΫΚβ≥Θ ΐ | 1.8ΓΝ10-5 | K1=4.3ΓΝ10-7 K2=5.6ΓΝ10-11 | 3.0ΓΝ10-8 |
ΜΊ¥πœ¬Ν–Έ ΧβΘΚ
Θ®1Θ©“ΜΑψ«ιΩωœ¬Θ§Β±Έ¬Ε»…ΐΗΏ ±Θ§Ka___(ΧνΓΑ‘ω¥σΓ±ΓΔΓΑΦθ–ΓΓ±ΜρΓΑ≤Μ±δΓ±)ΓΘ
Θ®2Θ©œ¬Ν–ΥΡ÷÷άκΉ”ΫαΚœ÷ Ή”ΡήΝΠ”…¥σΒΫ–ΓΒΡΥ≥–ρ «___(Χν–ρΚ≈)ΓΘ
a.CO32- b.ClO- c.CH3COO- d.HCO3-
Θ®3Θ©”Ο’τΝσΥ°œΓ Ά0.1mol/LΒΡ¥ΉΥαΘ§œ¬Ν–Ης Ϋ±μ ΨΒΡ ΐ÷ΒΥφΥ°ΝΩΒΡ‘ωΦ”Εχ‘ω¥σΒΡ «___(Χν–ρΚ≈)ΓΘ
a.![]() b.
b.![]()
c. ![]() d.
d.![]()
Θ®4Θ©≥ΘΈ¬œ¬Θ§0.1mol/LNaHCO3»ή“ΚΒΡpH¥σ”Ύ8Θ§‘ρ»ή“Κc(H2CO3)___c(CO32-)(ΧνΓΑΘΨΓΔΘΦΜρ=Γ±)Θ§‘≠“ρ «___ΓΘ
Θ®5Θ©25Γφ ±Θ§»τ≤βΒΟCH3COOH”κCH3COONaΒΡΜλΚœ»ή“ΚΒΡpH=6Θ§‘ρ»ή“Κ÷–ΘΚ
ΔΌ![]() =___(ΧνΓΑΨΪ»Ζ ΐ÷ΒΓ±Θ§œ¬Ά§)ΓΘ
=___(ΧνΓΑΨΪ»Ζ ΐ÷ΒΓ±Θ§œ¬Ά§)ΓΘ
ΔΎc(CH3COO-)-c(Na+)=___mol/LΓΘ