��Ŀ����
4��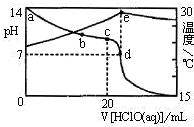 NaClO������ˮ�ľ�����ֽ��Ư�ס������ȣ�������ҽҩ��ҵ���������Ȱ���
NaClO������ˮ�ľ�����ֽ��Ư�ס������ȣ�������ҽҩ��ҵ���������Ȱ�����1��NaClO��Һ��ɱ��������ԭ��֮һ������Һ�д�����Ч�ȣ�HClO����д��NaClOˮ������ӷ���ʽ��ClO-+H2O
 HClO+OH-��
HClO+OH-����2��NaClO��Һ��ɱ��Ч������Һ��Ũ�ȡ��¶ȼ���Һ�д��ڵ����йأ�
�ٲ�ͬ�¶��£�500mL 0.1mol?L-1NaClO��Һ��1m3�ռ��ɱ��Ч�����±���ʾ��
| �¶�/�� | 20 | 30 | 40 | 80 |
| ʱ��/min | 10 | 10 | 10 | 10 |
| ɱ���� | 83% | 90% | 97% | 92% |
�������������NaClO��Һ����Ч������cd������ĸ����
a��NH4+ b��ClO- c��OH- d��SO32-
��3�������£���1.00mol?L-1��HClO����20.00mL 1.00mol?L-1NaOH��Һ�У���Һ��pH���¶������HClO����ı仯������ͼ��ʾ��
��a��b��c��d�ĵ��У�ˮ�ĵ���̶�������c������ĸ����
������˵������ȷ����AB������ĸ����
A��a��b��c��d�ĵ���Һ��d����Һ��ClO-�����ʵ������
B��c����Һ�У�c��Na+����c��ClO-����c��OH-����c��H+��
C��d����Һ�У�c��Na+��=c��ClO-��+c��HClO��
D��e�����Һ�¶��½�����Ҫԭ����HClO�������ȣ�
���� ��1��NaClOˮ��ˮ��HClO��Ϊ���淴Ӧ��
��2�������ϱ���֪��NaClO��Һ��40��ʱ��ɱ��Ч����������20��ʱ����֪�¶�Ӱ��ˮ�⣻
�ڼ���NaClO��Һ����Ч��������ˮ������䷢����Ӧ��
��3���ٽ�1.00mol?L-1��HClO����20.00mL 1.00mol?L-1NaOH��Һ��ǡ����ȫ��Ӧʱ�������δٽ�ˮ�ĵ��룻
��A��c��ǡ�÷�Ӧ��d��HClO������
B��c��ΪNaClO��Һ��ˮ���Լ��ԣ�
C��d��HClO������
D���кͷ�Ӧ���ȣ�������������ȡ�ˮ��Ҳ���ȣ���������Һ���¶Ƚϵͣ�
��� �⣺��1��NaClOˮ��ˮ��HClO��Ϊ���淴Ӧ��ˮ�����ӷ�ӦΪClO-+H2O HClO+OH-���ʴ�Ϊ��ClO-+H2O
HClO+OH-���ʴ�Ϊ��ClO-+H2O HClO+OH-��
HClO+OH-��
��2�������ϱ���֪��NaClO��Һ��40��ʱ��ɱ��Ч����������20��ʱ����֪�¶�Ӱ��ˮ�⣬��ԭ��Ϊ�����¶Ȼ�ٽ�ClO-��ˮ�⣬ʹHClOŨ������ɱ��Ч����ǿ���ʴ�Ϊ�������¶Ȼ�ٽ�ClO-��ˮ�⣬ʹHClOŨ������ɱ��Ч����ǿ��
�ڼ���NaClO��Һ����Ч��������ˮ������䷢����Ӧ��c����ˮ�⣬d���䷢��������ԭ��Ӧ��������HClO��Ũ�ȣ��ʴ�Ϊ��cd��
��3���ٽ�1.00mol?L-1��HClO����20.00mL 1.00mol?L-1NaOH��Һ��c��ǡ����ȫ��Ӧʱ�������δٽ�ˮ�ĵ��룬��c��ˮ�ĵ���̶���ʴ�Ϊ��c��
��A��c��ǡ�÷�Ӧ��d��HClO��������a��b��c��d�ĵ���Һ��d����Һ��ClO-�����ʵ������A��ȷ��
B��c��ΪNaClO��Һ��ˮ���Լ��ԣ������Һ�У�c��Na+����c��ClO-����c��OH-����c��H+������B��ȷ��
C���������غ��֪��c��c��Na+��=c��ClO-��+c��HClO����d��HClO��������d��c��Na+����c��ClO-��+c��HClO������C����
D���кͷ�Ӧ���ȣ��¶Ƚ�����Ҫ�������Һ���¶ȵ͵��£���D����
�ʴ�Ϊ��AB��
���� ���⿼������Ũ�ȴ�С�Ƚϡ�����ˮ��ȣ�ע�ظ�Ƶ����Ŀ��飬���շ�Ӧԭ��Ϊ���Ĺؼ������ط�����Ӧ�������Ŀ��飬��Ŀ�Ѷ��еȣ�

 �����ѧСѧ�꼶�νӵ������㽭��ѧ������ϵ�д�
�����ѧСѧ�꼶�νӵ������㽭��ѧ������ϵ�д� Сѧ�����ҵ���ϴ�ѧ������ϵ�д�
Сѧ�����ҵ���ϴ�ѧ������ϵ�д� ���Ž�����ٰθ��νӹ㶫���������ϵ�д�
���Ž�����ٰθ��νӹ㶫���������ϵ�д� �����������ҵ�������������ϵ�д�
�����������ҵ�������������ϵ�д�| A�� | �մ���Һ��ϡ���ᷴӦ�����ӷ�Ӧ����ʽ��HCO3-+H+�TCO2��+H2O | |
| B�� | Fe2O3��HI��Fe2O3+6H+�T2Fe3++3H2O | |
| C�� | NaHCO3��Һ�еμ���������ʯ��ˮ��2HCO3-+Ca2++2OH-�TCaCO3��+2H2O+CO32- | |
| D�� | Ũ�ռ���Һ�м�����Ƭ��Al+OH-+H2O�TAlO2-+H2�� |
| A�� | ����ˮ��Ӧ��Na+2H2O�TNa++2OH-+H2�� | |
| B�� | ��������Һ�������Һ��ϣ�SiO32-+2H+�TH2SiO3�� | |
| C�� | ��NaAlO2��Һ��ͨ�����CO2��Al��OH��3��CO2+AlO2-+2H2O�TAl��OH��3��+HCO3- | |
| D�� | ����������Һͨ������SO2��Ca2++2ClO-+SO2+H2O�TCaSO3��+2HClO |

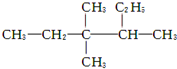 ������Ϊ3��3��4-��������
������Ϊ3��3��4-��������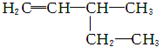 ������Ϊ3-��-1-��ϩ
������Ϊ3-��-1-��ϩ

 NH3•H2O+H+�������ӷ���ʽ��ʾ��������ʱ����NH4��2SO4����Һ�е���NaOH ��Һ����Һ�����ԣ���������Һ����Ũ�ȴ�С��ϵc��Na+��=c��NH3•H2O���������������������=����
NH3•H2O+H+�������ӷ���ʽ��ʾ��������ʱ����NH4��2SO4����Һ�е���NaOH ��Һ����Һ�����ԣ���������Һ����Ũ�ȴ�С��ϵc��Na+��=c��NH3•H2O���������������������=���� ��CԪ������������Ӧ��ˮ����ĵ���ʽ
��CԪ������������Ӧ��ˮ����ĵ���ʽ ��
�� ��
��