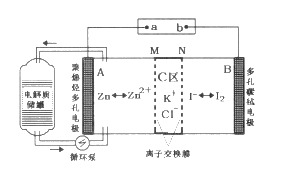
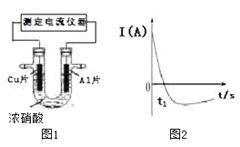
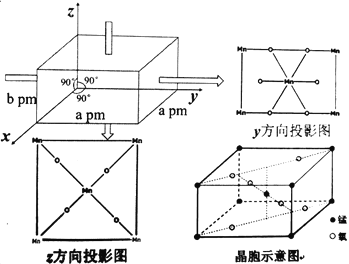
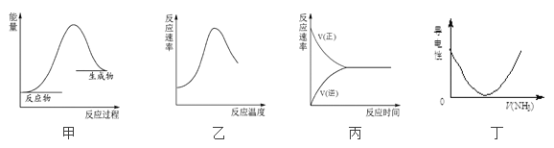
��Ŀ����
����Ŀ��25��ʱ������ƽ�ⳣ����
��ѧʽ | CH3COOH | H2CO3 | HClO |
����ƽ�ⳣ�� | 1.8��10-5 | K1=4.3��10-7 K2=5.6��10-11 | 3.0��10-8 |
��1��������ˮϡ��0.10mol/L�Ĵ��ᣬ�����и�ʽ��ʾ����ֵ��ˮ�������Ӷ��������_____��
A.![]() B.
B.![]() C.
C.![]() D.
D.![]()
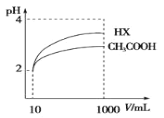
��2�����Ϊ10mLpH=2�Ĵ�����Һ��һԪ��HX�ֱ��ˮϡ����1000mL��ϡ����pH�仯��ͼ����HX�ĵ���ƽ�ⳣ��______(������������������������С����)�����ƽ�ⳣ����ϡ�ͺ�HX��Һ��ˮ���������c(H+)_____������Һ��ˮ���������c(H+)(������������������������С����)��
���𰸡�B ���� ����
��������
��1������ĵ��뷽��ʽΪ��CH3COOH![]() CH3COO-+H+����ˮϡ��CH3COOH��CH3COO-��H+��Ũ�Ⱦ���С��
CH3COO-+H+����ˮϡ��CH3COOH��CH3COO-��H+��Ũ�Ⱦ���С��
A����Ka= ��֪���¶Ȳ���Ka���䣬c(CH3COO-)��С����
��֪���¶Ȳ���Ka���䣬c(CH3COO-)��С���� ����
����![]() ����A����
����A����
B����Ka= ��֪���¶Ȳ���Ka���䣬c(H+)��С��
��֪���¶Ȳ���Ka���䣬c(H+)��С�� ����B��ȷ��
����B��ȷ��
C���¶Ȳ���KW���䣬c(H+)��С����![]() ��С��C����
����C����
D��KW=c(H+)��c(OH-)���¶Ȳ���KW���䣬c(H+)��С����c(OH-)��������![]() ��С��D����
����D����
�ʴ�ѡB��
��2��ϡ����ͬ�ı�����pH�仯��ĵ���̶ȴ���ͼ��֪��HX��pH�仯����HX�ĵ���̶ȴ���ƽ�ⳣ������ͼ��֪��ϡ�ͺ��������Ը�ǿ�����Ը�ǿ��������ˮ����ij̶ȸ��ʴ����У�ˮ���������c(H+)Ũ�ȸ�С���ʴ�Ϊ�����ڣ����ڡ�