��Ŀ����
17���ס��ҡ�����λͬѧ��ȡ�Ҵ���������Ũ�����Ϲ��ȵķ�����ȡ�Ҵ���������1����д���÷�Ӧ�Ļ�ѧ����ʽΪCH3COOH+CH3CH2OH
 CH3COOC2H5+H2O��
CH3COOC2H5+H2O��[װ�����]�ס��ҡ�����λͬѧ�ֱ����������������Һװ�ã�
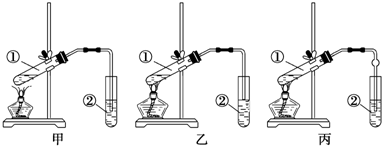
��2�����Ӽס�����λͬѧ��Ƶ�װ����ѡ��һ����Ϊʵ������ȡ����������װ�ã�ѡ���װ��Ӧ���ң���ס����ҡ�������ͬѧ����װ���еIJ����ܸij������θ���ܣ��������������⣬��һ��Ҫ�����Ƿ�ֹ������
[ʵ�鲽��]
A������ѡ���װ����װ���������Թܢ����ȼ���3mL��95%���Ҵ�������ҡ���»�������2mLŨ������ҡ�ȣ���ȴ���ټ���2mL�����
B�����Թ̶ܹ�������̨�ϣ�
C�����Թܢ��м���5mL����Na2CO3��Һ��
D���þƾ��ƶ��Թܢټ��ȣ�
E�����۲쵽�Թܢ�������������ʱֹͣʵ�飮
��3������A��װ��ʵ��װ�ã�������Ʒǰ��Ӧ���װ�������ԣ�
�Թܢ��б���Na2CO3��Һ�������dz�ȥ������Ҵ������������������ܽ�ȣ�
����Na2CO3��Һ������NaOH��Һ���棬��ԭ����CH3COOC2H5+NaOH��CH3COONa+CH3CH2OH���û�ѧ����ʽ˵������
���� ��1��������Ӧ�ı���Ϊ�����ǻ��������⣬�Ҵ������ᷴӦ��������������ˮ����Ϊ���淴Ӧ��
��2�����ݴֲ�Ʒ����������������Ҵ��Ļ�����и��Ե����ʣ��Լ����Ȳ����Ҳ�ܲ����������н��
��3��ʵ��ǰ��Ҫ���װ�õ������ԣ��ñ���̼������Һ���������������Ҵ��ܽ⣬̼���������ᷴӦ��ȥ���ᡢͬʱ���������������ܽ�ȣ���������������������Һ������ȫˮ�⣮
��� �⣺��1��������Ӧ�ı���Ϊ�����ǻ��������⣬�������Ҵ���Ũ���������¼��ȷ���������Ӧ��������������ˮ���÷�Ӧ����ʽΪ��CH3COOH+CH3CH2OH CH3COOC2H5+H2O��
CH3COOC2H5+H2O��
�ʴ�Ϊ��CH3COOH+CH3CH2OH CH3COOC2H5+H2O��
CH3COOC2H5+H2O��
��2�����������л����Ҵ������ᣬ����������ˮ���������������Ȳ����Ҳ�ܲ���������������װ����ȡ����ֹ������������ķ�������ͬѧ����װ���еIJ����ܸij������θ���ܣ��������������⣬���θ�������β��������ݻ��ϴ�Ҳ����ֹ���������ã�
�ʴ�Ϊ���ң���ֹ������
��3��ʵ��ǰ��Ҫ���װ�õ������ԣ�����ᵼ��ʵ��ʧ�ܣ�̼������Һ�ܹ������Ҵ����ܸ����ᷴӦ�к����ᣬ�����ŵ�������������ζ�������������ڱ���Na2CO3��Һ�е��ܽ�ȸ�С�������ڷֲ���������������ϡNaOH��Һ��Ӧ������������Ҳ����ϡNaOH��Һ����ȫˮ�⣬CH3COOC2H5+NaOH��CH3COONa+CH3CH2OH�������ϳ���ԭ��
�ʴ�Ϊ��װ�������ԣ���ȥ������Ҵ������������������ܽ�ȣ�CH3COOC2H5+NaOH��CH3COONa+CH3CH2OH��
���� ���⿼�������������Ʊ�����Ŀ�ѶȲ���ע�����ⱥ��̼������Һ�������Լ�������Ӧ�Ļ���������������ѧ���������������������Ӧ����ѧ֪ʶ���ʵ�������������

 һ������ϵ�д�
һ������ϵ�д� �Ƹ�С״Ԫ���ֳ������ϵ�д�
�Ƹ�С״Ԫ���ֳ������ϵ�д� �¸��̵�ѧϵ�д�
�¸��̵�ѧϵ�д� ����ͬѧһ����ʦȫ�źþ�ϵ�д�
����ͬѧһ����ʦȫ�źþ�ϵ�д�| A�� | 68.3% | B�� | 74.2% | C�� | 92.2% | D�� | 78.1% |
| A�� | ��ϩ��������ȼ��ʱ�ų���������˳����������ӻ��и���� | |
| B�� | �Ҷ����ͱ�����������ɫ����������ζ��Һ�壬���������ƻ�ױƷ | |
| C�� | ��֬�ڼ���������ˮ����ĸ��ͳ������������� | |
| D�� | ������ʳ��ƾ����˵��ۡ������ǡ��Ҵ��Ļ�ѧ�仯���� |
| A�� | λ�ڵڰ����ڣ��ڢ�A�� | |
| B�� | ����������Ӧˮ�������ǿ���� | |
| C�� | ��Ԫ�ص�ԭ�Ӱ뾶�ϴ��۷е�ܸ� | |
| D�� | ��������Ԫ�أ���Ȼ�粻���ڣ������з����� |
| A�� | NH3 | B�� | ��ϩ | C�� | ���� | D�� | ��ϩ |
| A�� | ͨʽ��ͬ�IJ�ͬ�л���һ������ͬϵ�� | |
| B�� | ������ͬ�����ŵ��л��ﲻһ����ͬϵ�� | |
| C�� | ����Ϊͬ���칹������ʲ����ܾ�����ͬ��ͨʽ | |
| D�� | ��Ϊͬ���칹�������֮����������һ����ͬ������ѧ����һ������ |
| A�� | Cu��OH��2��Һ�еμӴ��Cu��OH��2+2H+�TCu2++2H2O | |
| B�� | NaOH��Һ�м�����Ƭ��2Al+2 OH���T2AlO2��+3H2�� | |
| C�� | ��֪Fe2+���Ի�ԭC12��Br2��C12��������Br-����Br2���ڱ�״���£���10mL 0.1moL/LFeBr2��Һ��ͨ��22.4mL������2Fe2++2Br-+2C12�T2Fe3++Br2+4C1- | |
| D�� | NaHCO3��Һ��ϡ������Һ�ķ�Ӧ��HCO3-+H+�TH2O+CO2�� |
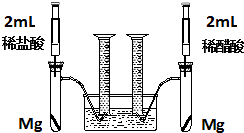 ʵ��һ��ij��ѧС��Ϊ�Ƚ�����ʹ�������ԣ����������ʵ�鷽����װ����ͼ��
ʵ��һ��ij��ѧС��Ϊ�Ƚ�����ʹ�������ԣ����������ʵ�鷽����װ����ͼ��