��Ŀ����
1��д�����з�Ӧ���Ȼ�ѧ����ʽ����1����25�桢101kPa�£�1g�״�ȼ������CO2��Һ̬ˮʱ����22.68kJ�����ʾ�״�ȼ���ȵ��Ȼ�ѧ����ʽΪCH3OH��l��+$\frac{3}{2}$O2��g����CO2��g��+2H2O��l����H=-725.76kJ•mol-1��
��2����������N2��O2��ȫ��Ӧ��ÿ����23g NO2��Ҫ����16.95kJ������N2��O2��Ӧ����NO2���Ȼ�ѧ����ʽΪN2��g��+2O2��g��=2NO2��g����H=+67.8kJ•mol-1��
��3����֪��1mol H-H����1molN-H����1molN��N���ֱ���Ҫ��������436kJ��391kJ��946kJ��
��N2��H2��Ӧ����NH3���Ȼ�ѧ����ʽΪN2��g��+3H2��g��=2NH3��g����H=-92kJ•mol-1��
��4����֪��N2H4��g��+O2��g���TN2��g��+2H2O��g����H=-543kJ/mol
N2��g��+2O2��g���T2NO2��g����H=+67.7kJ/mol
д��N2H4�Ͷ���������Ӧ���ɵ�������̬ˮ���Ȼ�ѧ����ʽΪ2N2H4��g��+2NO2 ��g���T3N2��g��+4 H2O��g����H=-1135.7kJ•mol-1��
���� ��1��ȼ������1mol��ȼ����ȫȼ�������ȶ�������ʱ�ų�����������25�桢101kPa�£�1g�״���CH3OH��ȼ������CO2��Һ̬ˮʱ����22.68kJ��1mol�״���ȫȼ�����ɶ�����̼��Һ̬ˮ����725.8KJ��
��2��������֪�������Ӧ�ȣ��ٸ����Ȼ�ѧ����ʽ����д������
��3����ѧ��Ӧ�У���ѧ�����������������γ��»�ѧ���ų����������ݷ���ʽ����ֱ����պͷų����������Դ˼��㷴Ӧ�Ȳ��ж����Ȼ��Ƿ��ȣ�
��4���ݸ�˹���ɣ�����֪�Ȼ�ѧ����ʽ�����ʵ���ϵ�����мӼ�����Ŀ���Ȼ�ѧ����ʽ����Ӧ��Ҳ������Ӧ��ϵ����������Ӧ�ļӼ���
��� �⣺��1����25�桢101kPa�£�1g�״���CH3OH��ȼ������CO2��Һ̬ˮʱ����22.68kJ��1mol�״���ȫȼ�����ɶ�����̼��Һ̬ˮ����Ϊ22.68kJ��32=725.76KJ�����Լ״�ȼ���ȵ��Ȼ�ѧ����ʽΪCH3OH��l��+$\frac{3}{2}$O2��g���TCO2��g��+2H2O��l����H=-725.76 kJ•mol-1��
�ʴ�Ϊ��CH3OH��l��+$\frac{3}{2}$O2��g���TCO2��g��+2H2O��l����H=-725.76 kJ•mol-1��
��2��������N2��O2��ȫ��Ӧ��ÿ����23��NO2��Ҫ����16.95kJ����������ÿ����92��NO2��Ҫ����67.8kJ������
���Ȼ�ѧ����ʽΪN2��g��+2O2��g��=2NO2��g����H=+67.8kJ•mol-1��
�ʴ�Ϊ��N2��g��+2O2��g��=2NO2��g����H=+67.8kJ•mol-1��
��3���ڷ�ӦN2+3H2?2NH3�У�����3molH-H����1molN��N�������յ�����Ϊ3��436kJ+946kJ=2254kJ������2molNH3�����γ�6molN-H�����ų�������Ϊ6��391kJ=2346kJ�����յ������٣��ų��������࣬�÷�ӦΪ���ȷ�Ӧ���ų�������Ϊ2346kJ-2254kJ=92kJ��N2��H2��Ӧ����NH3���Ȼ�ѧ����ʽΪ��N2��g��+3H2��g��?2NH3��g����H=-92kJ•mol-1��
�ʴ�Ϊ��N2��g��+3H2��g��?2NH3��g����H=-92kJ•mol-1��
��4����N2��g��+2O2��g��=2NO2��g����H=+67.7kJ/mol
��N2H4��g��+O2��g���TN2��g��+2H2O ��g������H=-534kJ•mol-1
�ɸ�˹���ɣ��ڡ�2-�ٵ� 2N2H4��g��+2NO2 ��g���T3N2��g��+4 H2O��g����H=-1135.7kJ•mol-1
�ʴ�Ϊ��2N2H4��g��+2NO2 ��g���T3N2��g��+4 H2O��g����H=-1135.7kJ•mol-1��
���� ���⿼�����Ȼ�ѧ����ʽ����д��������Ҫ��ȼ���ȸ���������������Ŀ�ϼ����ջ����ǽ���ؼ���

��101kPaʱ��2C��s��+O2��g���T2CO��g������H=-221kJ/mol
��ϡ��Һ�У�H+��aq��+OH-��aq���TH2O����H=-57.3kJ/mol
���н�����ȷ���ǣ�������
| A�� | ̼��ȼ���ȴ���-110.5 kJ/mol | |
| B�� | �ٵķ�Ӧ��Ϊ-221 kJ/mol | |
| C�� | ϡ������ϡNaOH��Һ��Ӧ���к���Ϊ-114.6 kJ/mol | |
| D�� | ϡ������ϡNaOH��Һ��Ӧ����1molˮ���ų�57.3 kJ���� |
��1���÷�Ӧ���Ȼ�ѧ����ʽΪN2H4��l��+2H2O2��l���TN2��g��+4H2O��g����H=-641.625kJ/mol��
��2������֪��H2O��g����H2O��L����H=-44KJ/moL����16gҺ̬����Һ̬˫��ˮ��Ӧ����Һ̬ˮʱ�ų���������408.8kJ��
��3���˷�ӦӦ���ڻ���ƽ������ͷŴ����ȺͿ��ٲ������������⣬����һ���ܴ���ŵ��������ﲻ����Ⱦ������
��4����֪1g������ȫȼ������Һ̬ˮʱ�ų�����121kJ������������������
| O=O��g�� | H-H��g�� | H-O��g�� | |
| 1mol��ѧ������ʱ��Ҫ���յ�����/kJ | 496 | X | 463 |

| A�� | ��t1=15 s������A��Ũ�ȱ仯��ʾ��t0��t1�ε�ƽ����Ӧ����Ϊ0.004mol•L-1•s-1 | |
| B�� | t4��t5�θı������һ��Ϊ����ѹǿ | |
| C�� | ���������ݻ�Ϊ2 L��B����ʼ���ʵ���Ϊ0.02 mol | |
| D�� | t5��t6�Σ�������A�����ʵ���������0.06 mol�����˹����������������Ƚ�������Ϊa kJ���÷�Ӧ���Ȼ�ѧ����ʽΪ��3A��g��?B��g��+2C��g����H=+50a kJ•mol-1 |
| A�� | HCl��NaOH��Ӧ���к��ȡ�H=-57.3kJ•mol-1����1mol H2SO4��������ˮ��Ӧ���ͷŵ�����Ϊ114.6kJ | |
| B�� | ��֪��1��105Pa��298K�����£�2mol����ȼ������ˮ�������ų�484 kJ���������У�H2O��g���TH2��g��+$\frac{1}{2}$O2��g����H=+242kJ/mol | |
| C�� | ȼ�ϵ���н��״�����ת��Ϊ�������Ȼ�ѧ����ʽ�ǣ�CH3OH��g��+$\frac{1}{2}$O2��g���TCO2��g��+2H2��g����H=-192.9kJ•mol-1����CH3OH��g����ȼ����Ϊ192.9kJ•mol-1 | |
| D�� | ��Ҫ���Ȳ��ܷ����ķ�Ӧһ�������ȷ�Ӧ |
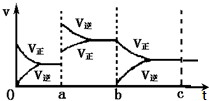 ��ͼ��ʾ���ܱ������з�Ӧ��2SO2+O2?2SO3+Q�ﵽƽ��ʱ�����������ı������Ӧ�ٶȺͻ�ѧƽ��ı仯�����ab�����иı���������������£�bc������ab������Ȼ�ѧƽ�ⳣ�����䣮���������С�����䡱��
��ͼ��ʾ���ܱ������з�Ӧ��2SO2+O2?2SO3+Q�ﵽƽ��ʱ�����������ı������Ӧ�ٶȺͻ�ѧƽ��ı仯�����ab�����иı���������������£�bc������ab������Ȼ�ѧƽ�ⳣ�����䣮���������С�����䡱��