ΧβΡΩΡΎ»ί
ΓΨΧβΡΩΓΩΡ≥![]() ΒΡ»ή“ΚXΘ§Τδ÷–Ω…ΡήΚ§”–
ΒΡ»ή“ΚXΘ§Τδ÷–Ω…ΡήΚ§”–![]() ΓΔ
ΓΔ![]() ΓΔ
ΓΔ![]() ΓΔ
ΓΔ![]() ΓΔ
ΓΔ![]() ΓΔ
ΓΔ![]() ΓΔ
ΓΔ![]() ΓΔ
ΓΔ![]() ΓΔ
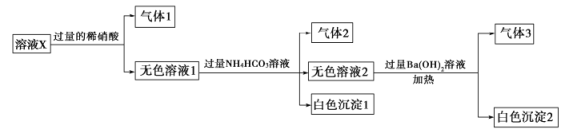
ΓΔ![]() ÷–ΒΡ“Μ÷÷ΜρΦΗ÷÷Θ§»Γ200 mLΗΟ»ή“ΚΫχ–– Β―ιΘ§Τδœ÷œσΦΑΉΣΜ·»γΆΦΘ°
÷–ΒΡ“Μ÷÷ΜρΦΗ÷÷Θ§»Γ200 mLΗΟ»ή“ΚΫχ–– Β―ιΘ§Τδœ÷œσΦΑΉΣΜ·»γΆΦΘ°
“―÷ΣΘΚΖ¥”ΠΙΐ≥Χ÷–”–“Μ÷÷ΤχΧε «ΚλΉΊ…ΪΘ°
«κΜΊ¥πœ¬Ν–Έ ΧβΘΚ
Θ®1Θ©Ϋω”…«ΩΥα–‘ΧθΦΰ±ψΩ…≈–Εœ»ή“ΚX÷–“ΜΕ®≤Μ¥φ‘ΎΒΡάκΉ””–________Θ°
Θ®2Θ©»ή“ΚX÷–Θ§ΙΊ”Ύ![]() ΒΡ≈–Εœ“ΜΕ®’ΐ»ΖΒΡ «________Θ®ΧνΉ÷ΡΗΘ©Θ°
ΒΡ≈–Εœ“ΜΕ®’ΐ»ΖΒΡ «________Θ®ΧνΉ÷ΡΗΘ©Θ°
aΘ°“ΜΕ®”– bΘ°“ΜΕ®ΟΜ”– cΘ°Ω…Ρή”–
Θ®3Θ©ΔΌ÷–≤ζ…ζΤχΧεAΒΡάκΉ”ΖΫ≥Χ ΫΈΣ_____________________________________________________Θ°
Θ®4Θ©ΔΏ÷–…ζ≥…≥ΝΒμKΒΡάκΉ”ΖΫ≥Χ ΫΈΣ_____________________________________________________Θ°
Θ®5Θ©»ή“ΚX÷–≤ΜΡή»ΖΕ®ΒΡάκΉ” «_____________________”Ο Β―ι÷ΛΟςΗΟάκΉ”“ΜΕ®≤Μ¥φ‘ΎΒΡΜ·―ßΖΫΖ®ΈΣ______________________________________________________________________________________Θ°
Θ®6Θ©»τ Β―ι≤βΕ®AΘ°FΘ°KΨυΈΣ![]() lΘ§ ‘»ΖΕ®≥ΝΒμCΦΑΤδΈο÷ ΒΡΝΩΖΕΈßΘΚ_________Θ°
lΘ§ ‘»ΖΕ®≥ΝΒμCΦΑΤδΈο÷ ΒΡΝΩΖΕΈßΘΚ_________Θ°
ΓΨ¥πΑΗΓΩ![]() ΓΔ
ΓΔ![]() b
b ![]()
![]()
![]() ΓûÅΌΝΩ»ή“ΚX”Ύ ‘Ιή÷–Θ§Φ”»κΦΗΒΈKSCN»ή“ΚΘ§»ή“Κ≤Μ±δΚλ…ΪΘ§ΥΒΟςΈό
ΓûÅΌΝΩ»ή“ΚX”Ύ ‘Ιή÷–Θ§Φ”»κΦΗΒΈKSCN»ή“ΚΘ§»ή“Κ≤Μ±δΚλ…ΪΘ§ΥΒΟςΈό![]() Θ®ΤδΥϊ¥πΑΗΚœάμ“≤Ω…Θ© ≥ΝΒμΈΣ
Θ®ΤδΥϊ¥πΑΗΚœάμ“≤Ω…Θ© ≥ΝΒμΈΣ![]()
![]()
ΓΨΫβΈωΓΩ
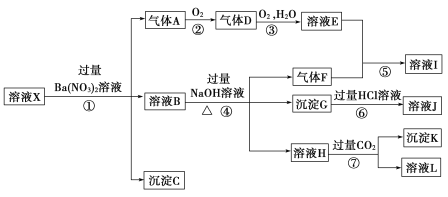
”…Ζ¥”ΠΔΌΦ”»κΒΡBa(NO3)2»ή“ΚΦΑpH= 1Θ§ΫαΚœ»ή“Κ÷–Ω…ΡήΚ§”–ΒΡάκΉ”Β»–≈œΔΩ…≈–ΕœΤχΧεAΈΣNOΘ§ΤχΧεDΈΣNO2Θ§»ή“ΚEΈΣHNO3Θ§“ρ¥Υ»ή“Κ÷–Κ§”–ΜΙ‘≠–‘ΒΡάκΉ”Θ§Φ¥Κ§”–Fe2+ΘΜ”…Ζ¥”ΠΔήΦ”»κΙΐΝΩNaOH»ή“ΚΦΑ»ή“Κ÷–Ω…Ρή¥φ‘ΎΒΡάκΉ”≈–ΕœΤχΧεFΈΣNH3Θ§»ή“ΚIΈΣNH4NO3Θ§»ή“Κ÷–Κ§”–NH4+; pH= 1ΒΡ»ή“Κ÷–≤ΜΩ…ΡήΚ§”–CO32-ΓΔSiO32-, Ι ≥ΝΒμCΈΣBaSO4;ΗυΨίΆΦ ΨΙΊœΒΦΑΩ…Ρή¥φ‘ΎΒΡάκΉ”Ω…÷Σ≥ΝΒμGΈΣFe(OH)3Θ§»ή“ΚJΈΣFeCl3;≥ΝΒμKΈΣAl(OH)3ΓΘ
Θ®1Θ©”…«ΩΥα–‘ΧθΦΰΦ¥Ω…≈–Εœ»ή“ΚX÷–“ΜΕ®≤Μ¥φ‘Ύ»θΥαΗυάκΉ”Θ§Υυ“‘≤Μ¥φ‘ΎΒΡάκΉ””–CO32-ΓΔSiO32-ΘΜ
Θ®2Θ©«ΩΥα–‘»ή“Κ÷–œθΥαΗυάκΉ”ΨΏ”–«Ω―θΜ·–‘Θ§»γΙϊ¥φ‘ΎœθΥαΗυάκΉ”Θ§‘ρ≤Μ¥φ‘Ύ―«ΧζάκΉ”Θ§Φ”»κœθΥα±Β»ή“Κ ±≤ΜΡή≤ζ…ζΤχΧεΘ§Υυ“‘»ή“ΚX÷–“ΜΕ®ΟΜ”–NO3-Θ§
¥πΑΗ―ΓbΘΜ
Θ®3Θ©―«ΧζάκΉ”ΚΆœθΥαΖ¥”Π…ζ≥…ΧζάκΉ”ΓΔ“Μ―θΜ·ΒΣΚΆΥ°Θ§άκΉ”Ζ¥”ΠΖΫ≥Χ ΫΈΣΘΚ3Fe2++NO3-+4H+=3Fe3++NOΓϋ+2H2OΘΜ
Θ®4Θ©ΔΏ÷–…ζ≥…≥ΝΒμKΈΣAl(OH)3Θ§Ζ¥”ΠΒΡάκΉ”ΖΫ≥Χ ΫΈΣAlO2-+CO2+2H2O= Al(OH)3Γΐ+HCO3-ΘΜ
(5)…ζ≥…Fe(OH)3ΒΡFe3+Ω…Ρήά¥Ή‘”ΎFe2+±ΜœθΥα―θΜ·Θ§“≤Ω…Ρή «‘≠ά¥»ή“Κ÷–ΨΆ¥φ‘ΎFe3+Θ§Ι ≤ΜΡή»ΖΕ®»ή“ΚX÷– «Ζώ¥φ‘ΎFe3+Θ§Ω…”ΟKSCN»ή“ΚΦλ―ι»ή“ΚX÷– «Ζώ¥φ‘ΎFe3+ΘΜ
(6) NOΓΔNH3ΓΔAl(OH)3 ΗςΈΣ0.01 molΘ§‘ρΥΒΟς»ή“ΚX÷–”–0.03molFe2+ΓΔ 0.01 mol NH4+ΓΔ0.01 mol Al3+ΓΘ »τ»ή“ΚX÷–≤Μ¥φ‘ΎFe3+,”……œ ωΆΤΕœΩ…÷Σ»ή“Κ÷–“ΜΕ®¥φ‘ΎAl3+ΓΔNH4+ΓΔFe2+ΓΔ SO42-Θ§”…»ή“Κ÷–ΒΡΒγΚ… ΊΚψΩ…÷Σ3n(Al3+) + n(NH4+)+ 2n(Fe2+)+ n(H+)= 2n(SO42-)Θ§Ι n(SO42-)= ![]() (0.03mol+0.01mol + 0.06 mol+ 0.02 mol)= 0.06 mol,»τ»ή“Κ÷–Κ§”–Fe3+Θ§‘ρn(SO42- )>0.06 molΓΘ
(0.03mol+0.01mol + 0.06 mol+ 0.02 mol)= 0.06 mol,»τ»ή“Κ÷–Κ§”–Fe3+Θ§‘ρn(SO42- )>0.06 molΓΘ

ΓΨΧβΡΩΓΩ“ΜΕ®Έ¬Ε» ±Θ§œρ2.0LΚψ»ίΟή±’»ίΤς÷–≥δ»κ2molSO2ΚΆ1mol O2Θ§ΖΔ…ζΖ¥”ΠΘΚ2SO2(g)ΘΪO2(g)![]() 2SO3(g)ΓΘΨ≠Ιΐ“ΜΕΈ ±ΦδΚσ¥οΒΫΤΫΚβΓΘΖ¥”ΠΙΐ≥Χ÷–≤βΕ®ΒΡ≤ΩΖ÷ ΐΨίΦϊœ¬±μΘΚ
2SO3(g)ΓΘΨ≠Ιΐ“ΜΕΈ ±ΦδΚσ¥οΒΫΤΫΚβΓΘΖ¥”ΠΙΐ≥Χ÷–≤βΕ®ΒΡ≤ΩΖ÷ ΐΨίΦϊœ¬±μΘΚ
t/s | 0 | 2 | 4 | 6 | 8 |
n(SO3)/mol | 0 | 0.8 | 1.4 | 1.8 | 1.8 |
œ¬Ν–ΥΒΖ®’ΐ»ΖΒΡ «Θ® Θ©
A.Ζ¥”Π‘Ύ«Α2sΒΡΤΫΨυΥΌ¬ v(O2)ΘΫ0.2molΓΛLΘ≠1ΓΛsΘ≠1
B.±Θ≥÷ΤδΥϊΧθΦΰ≤Μ±δΘ§ΧεΜΐ―ΙΥθΒΫ1.0LΘ§ΤΫΚβ≥Θ ΐΫΪ‘ω¥σ
C.œύΆ§Έ¬Ε»œ¬Θ§Τπ Φ ±œρ»ίΤς÷–≥δ»κ4molSO3Θ§¥οΒΫΤΫΚβ ±Θ§SO3ΒΡΉΣΜ·¬ ¥σ”Ύ10%
D.±Θ≥÷Έ¬Ε»≤Μ±δœρΗΟ»ίΤς÷–‘Ό≥δ»κ2molSO2ΓΔ1molO2Θ§Ζ¥”Π¥οΒΫ–¬ΤΫΚβ ±![]() ‘ω¥σ
‘ω¥σ