��Ŀ����
����Ŀ��25��ʱ���ֱ���20��00mL 0.10mol��L-1�İ�ˮ���������Һ�еμ�0.10mol��L-1������ᣬ��ҺpH��������������ı仯��ϵ��ͼ��ʾ������˵����ȷ����
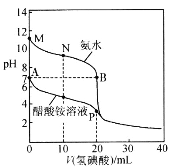
A.A����B����ˮ���������������Ũ��A�����B��
B.M�㣬c(NH3��H2O)>c(OH-)>c(H+)>c(NH4+)
C.N�㣬c(I-)=c(NH3��H2O)+c(NH4+)
D.P�㣬c(H+)+c(CH3COOH)=c(OH-)+c(I-)+c(NH3��H2O)
���𰸡�AD
��������
A��A��Ϊ�������Һ�������Ϊ���������Σ��ٽ�ˮ�ĵ��룬B��Ϊ�⻯��백ˮ�Ļ��Һ�������ƣ�Ҳ���ٽ�ˮ�ĵ��룬��ˮ���������������Ũ��A�����B�㣬A˵����ȷ��
B��M��Ϊ��ˮ����Һ�Լ��ԣ�c(NH3��H2O)>c(OH-)>c(NH4+)>c(H+)��B˵������
C��N��Ϊ�⻯��백ˮ�Ļ��Һ�������ʵ�����ȣ����������غ㣬2c(I-)=c(NH3��H2O)+c(NH4+)��C˵������
D��P��Ϊ�⻯������Ļ��Һ�������ʵ�����ȣ����ݵ���غ㣬c(NH4+)+c(H+)= c(OH-)+c(I-)+ c(CH3COO-)�����������غ㣬c(NH3��H2O)+c(NH4+)=c(I-)=c(CH3COOH)+c(CH3COO-)������c(NH4+)=c(I-)-c(NH3��H2O)��c(CH3COO-)=c(CH3COOH)- c(I-)�������غ�ĵ�ʽ�����ɵõ�c(H+)+c(CH3COOH)=c(OH-)+c(I-)+c(NH3��H2O)��D˵����ȷ��
��ΪAD��

 �����Ƹ���ʦ����ϵ�д�
�����Ƹ���ʦ����ϵ�д� ��ͨ����ͬ����ϰ��ϵ�д�
��ͨ����ͬ����ϰ��ϵ�д�����Ŀ������ʵ���о��к���ɫ����������Աȷ������ý��۲���ȷ���ǣ� ��
|
|
|
�� | �� | �� |
A.�ɢ��еĺ���ɫ���壬�ƶϲ���������һ���ǻ������
B.����ɫ���岻�ܱ�������ľ̿��Ũ���ᷢ���˷�Ӧ
C.�ɢ�˵��Ũ������лӷ��ԣ����ɵĺ���ɫ����Ϊ��ԭ����
D.�۵���������м���CO2���ɴ�˵��ľ̿һ����Ũ���ᷢ���˷�Ӧ
����Ŀ����������������������Ʒ��������̨������Ȧ����ʽ���У� ����ƿ ����ʽ�ζ��ܺͼ�ʽ�ζ��� ���ձ� �ݲ����� ��ͷ�ι� ����ƽ�������룩 ����ֽ ����Ͳ �����©����
��������ҩƷ����A��NaOH���� ��B����NaOH��Һ��C��δ֪Ũ������ ��D������ˮ��E��̼������Һ
������������ѧ��ʵ�飬�ش��������⣺
��1������ʱ��Ӧѡ�õ�����������_________________________�����ţ���
��2������һ�����ʵ���Ũ�ȵ�NaOH��Һʱ����ȱ�ٵ�������__________��
��3��������к͵ζ�ʱ����ȱ�����Լ���____________________��
��4�������к͵ζ�ʱ���������ý�Ҫʢ����Һ������ϴ�����������е�______�����ţ����ף���ʽ�ζ��� �ң���ʽ�ζ��� ������ƿ
��5���ñ���NaOH��Һ�ζ�δ֪Ũ�ȵ����ᣬѡ�÷�̪��Ϊָʾ������ɲⶨ���ƫ�ߵ�ԭ�������______________��
A�����Ʊ���Һ��NaOH�л���Na2CO3����
B���ζ��յ����ʱ�����ӵζ��ܵĿ̶ȣ�����������ȷ
C��ʢװδ֪Һ����ƿ������ˮϴ����δ��δ֪Һ��ϴ
D���ζ�ǰ�����ݣ��ζ���������ʧ��
II. �״���CH3OH����һ�ֿ�������Դ�����й㷺�Ŀ�����Ӧ��ǰ����
��1����ҵ��һ������������ַ�Ӧ�ϳɼ״���
��ӦI��CO2(g)+3H2(g)![]() CH3OH(g)+H2O(g) ��H1
CH3OH(g)+H2O(g) ��H1
��ӦII��CO(g)+2H2(g)![]() CH3OH(g) ��H2
CH3OH(g) ��H2
�ٷ�ӦI��ƽ�ⳣ������ʽΪK=________________________
���±����������Ƿ�Ӧ���ڲ�ͬ�¶��µĻ�ѧƽ�ⳣ��(K)
�¶� | 250�� | 300�� | 350�� |
K | 2.041 | 0.270 | 0.012 |
�ɱ��������ж���H2______0����S______0 (��������������������������)��
��ij�¶��£���2 mol CO��6 mol H2����2L���ܱ������У���ַ�Ӧ���ﵽƽ����c(CO)=0.2 mol��L-1����CO��ת����Ϊ________��
��2����֪�ڳ��³�ѹ�£�ijʵ��С�����ݼ״�ȼ�յķ�Ӧԭ�����������ͼ��ʾ�ĵ��װ�ã�

��2CH3OH(l)+3O2(g)=2CO2(g)+4H2O(g) ��H ��-1275.6 kJ��mol-1
��H2O(g)��H2O(l) ��H ��-44.0 kJ��mol-1
��д����ʾ�״�ȼ���ȵ��Ȼ�ѧ����ʽ____________________��
��д����ͼ���װ���и�����Ӧʽ��________________________________��





