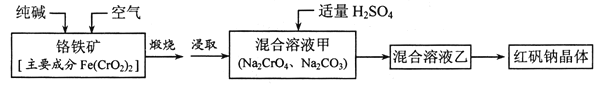
ƒøƒ⁄»›
°æƒø°ø2019ƒÍ≈µ±¥∂˚ªØ—ßΩ± ⁄”Ë‘º∫≤°§π≈µ¬“¡ƒ…∑Ú°¢ÀπÃπ¿˚°§ª›Õ¢∂Úƒ∑∫Õº™“∞’√»˝Œªø∆—ߺ“£¨“‘±Ì’√À˚√«‘⁄ÔƵÁ≥ÿ¡Ï”ÚÀ˘◊ˆ≥ˆµƒæfi¥Ûπ±œ◊°£![]() °¢
°¢![]() ≥£”√◊˜ÔÆ¿Î◊”µÁ≥ÿµƒ’˝º´≤ƒ¡œ£¨«Îªÿ¥œ¬¡–Œ £∫
≥£”√◊˜ÔÆ¿Î◊”µÁ≥ÿµƒ’˝º´≤ƒ¡œ£¨«Îªÿ¥œ¬¡–Œ £∫
(1)ª˘Ã¨ÔÆ‘≠◊”µƒ◊Ó∏flƒ‹º∂µƒµÁ◊”‘∆–Œ◊¥ «________£ªª˘Ã¨¡◊‘≠◊””–________∏ˆŒ¥≥…∂‘µÁ◊”£ªª˘Ã¨Ã˙‘≠◊”∫ÀÕ‚µÁ◊”≈≈≤º ΩŒ™________°£
(2)![]() ÷–
÷–![]() µƒ≈‰Œª ˝Œ™4£¨≈‰ÃÂ÷–Nµƒ‘”ªØ∑Ω ΩŒ™________£¨∏√≈‰¿Î◊”÷–∏˜‘™Àÿµƒµ⁄“ªµÁ¿Îƒ‹”…–°µΩ¥ÛµƒÀ≥–ÚŒ™________(”√‘™Àÿ∑˚∫≈±Ì æ)°£
µƒ≈‰Œª ˝Œ™4£¨≈‰ÃÂ÷–Nµƒ‘”ªØ∑Ω ΩŒ™________£¨∏√≈‰¿Î◊”÷–∏˜‘™Àÿµƒµ⁄“ªµÁ¿Îƒ‹”…–°µΩ¥ÛµƒÀ≥–ÚŒ™________(”√‘™Àÿ∑˚∫≈±Ì æ)°£
(3)![]() ‘⁄ÀÆ÷–“◊±ªªπ‘≠≥…
‘⁄ÀÆ÷–“◊±ªªπ‘≠≥…![]() £¨∂¯‘⁄∞±ÀÆ÷–ø…Œ»∂®¥Ê‘⁄£¨∆‰‘≠“ÚŒ™________°£
£¨∂¯‘⁄∞±ÀÆ÷–ø…Œ»∂®¥Ê‘⁄£¨∆‰‘≠“ÚŒ™________°£
(4)![]() Ù”⁄ºÚµ•¡◊À·—Œ£¨∂¯÷±¡¥µƒ∂‡¡◊À·—Œ‘Ú «“ª÷÷∏¥‘”¡◊À·—Œ£¨»Á£∫Ωπ¡◊À·ƒ∆°¢»˝¡◊À·ƒ∆µ»°£Ωπ¡◊À·∏˘¿Î◊”°¢»˝¡◊À·∏˘¿Î◊”»Áœ¬ÕºÀ˘ æ£∫
Ù”⁄ºÚµ•¡◊À·—Œ£¨∂¯÷±¡¥µƒ∂‡¡◊À·—Œ‘Ú «“ª÷÷∏¥‘”¡◊À·—Œ£¨»Á£∫Ωπ¡◊À·ƒ∆°¢»˝¡◊À·ƒ∆µ»°£Ωπ¡◊À·∏˘¿Î◊”°¢»˝¡◊À·∏˘¿Î◊”»Áœ¬ÕºÀ˘ æ£∫

’‚¿‡¡◊À·∏˘¿Î◊”µƒªØ—ß Ωø…”√Õ® Ω±Ì æŒ™________(”√n¥˙±ÌP‘≠◊” ˝)°£
(5)Ó‹¿∂æßÃÂΩ·ππ»Áœ¬Õº£¨∏√¡¢∑Ωæß∞˚”…4∏ˆ¢Ò–Õ∫Õ4∏ˆ¢Ú–Õ–°¡¢∑ΩÃÂππ≥…°£æßÃÂ÷–![]() ’ºæ›
’ºæ›![]() –Œ≥…µƒ________(ÃÓ°∞Àƒ√ÊÃÂø’œ∂°±ªÚ°∞∞À√ÊÃÂø’œ∂°±)£ªÓ‹¿∂æßõƒ√‹∂»Œ™___________
–Œ≥…µƒ________(ÃÓ°∞Àƒ√ÊÃÂø’œ∂°±ªÚ°∞∞À√ÊÃÂø’œ∂°±)£ªÓ‹¿∂æßõƒ√‹∂»Œ™___________![]() (¡–≥ˆº∆À„ Ω£¨”√
(¡–≥ˆº∆À„ Ω£¨”√![]() ±Ì æ∞¢∑¸º”µ¬¬fi≥£ ˝µƒ÷µ)°£
±Ì æ∞¢∑¸º”µ¬¬fi≥£ ˝µƒ÷µ)°£

°æ¥∞∏°ø«Ú–Œ 3 ![]() £®ªÚ
£®ªÚ![]() £©
£© ![]()
![]()
![]() ø…”Î
ø…”Î![]() –Œ≥…ΩœŒ»∂®µƒ≈‰∫œŒÔ
–Œ≥…ΩœŒ»∂®µƒ≈‰∫œŒÔ ![]() ∞À√ÊÃÂø’œ∂
∞À√ÊÃÂø’œ∂ 
°æΩ‚Œˆ°ø
(1)ÔÆ «3∫≈‘™Àÿ£¨∫ÀÕ‚µÁ◊”≈≈≤ºŒ™1s22s1£¨◊Ó∏flƒ‹º∂ «2s£¨∆‰µÁ◊”‘∆–Œ◊¥ ««Ú–Œ£ª¡◊ «15∫≈‘™Àÿ£¨∫ÀÕ‚µÁ◊”≈≈≤º ΩŒ™1s22s22p63s23p3£¨∆‰Œ¥≥…∂‘µÁ◊” ˝ «3pƒ‹º∂…œµƒ3∏ˆµÁ◊”£ªÃ˙ «26∫≈‘™Àÿ£¨C∏˘æ›∫ÀÕ‚µÁ◊”≈≈≤ºπʬ…ø…µ√∆‰ª˘Ã¨Ã˙‘≠◊”∫ÀÕ‚µÁ◊”≈≈≤º Ω1s22s22p63s23p63d64s2ªÚ[Ar]3d64s2£ª
π ¥∞∏Œ™£∫«Ú–Œ£ª3£ª1s22s22p63s23p63d64s2ªÚ[Ar]3d64s2£ª
(2)NO3£≠÷–º€≤„µÁ◊”∂‘ ˝Œ™5+0+12=3£¨π Œ™sp2‘”ªØ£ª“ª∞„«Èøˆœ¬∑«Ω Ù–‘‘Ω«øµ⁄“ªµÁ¿Îƒ‹‘Ω¥Û£¨µ´”…”⁄N‘≠◊”÷–◊ÓÕ‚≤„Œ™∞Î≥‰¬˙◊¥Ã¨£¨±»ΩœŒ»∂®£¨π µ⁄“ªµÁ¿Îƒ‹¥Û”⁄O£¨À˘“‘µ⁄“ªµÁ¿Îƒ‹”…–°µΩ¥ÛµƒÀ≥–ÚŒ™![]() £ª
£ª
π ¥∞∏Œ™£∫sp2£ª![]() £ª
£ª
(3)![]() ø…”Î
ø…”Î![]() –Œ≥…ΩœŒ»∂®µƒ≈‰∫œŒÔ£¨π
–Œ≥…ΩœŒ»∂®µƒ≈‰∫œŒÔ£¨π ![]() ‘⁄ÀÆ÷–“◊±ªªπ‘≠≥…
‘⁄ÀÆ÷–“◊±ªªπ‘≠≥…![]() £¨∂¯‘⁄∞±ÀÆ÷–ø…Œ»∂®¥Ê‘⁄£ª
£¨∂¯‘⁄∞±ÀÆ÷–ø…Œ»∂®¥Ê‘⁄£ª
(4)ø…“‘∏˘æ›¡◊À·∏˘°¢Ωπ¡◊À·∏˘°¢»˝¡◊À·∏˘µƒªØ—ß ΩÕ∆µº£∫PO43-°¢P2O74-°¢P3O105-¡◊‘≠◊”µƒ±‰ªØπʬ…Œ™£∫1°¢2°¢3°¢4£¨n—ı‘≠◊”µƒ±‰ªØπʬ…Œ™£∫4°¢7°¢10°¢3n+1À·∏˘À˘¥¯µÁ∫… ˝µƒ±‰ªØπʬ…Œ™£∫3°¢4°¢5°¢n+2£¨’‚¿‡¡◊À·∏˘¿Î◊”µƒªØ—ß Ωø…”√Õ® Ω![]() ¿¥±Ì 棪
¿¥±Ì 棪
(5£©∏˘æ›Ó‹¿∂æßÃÂæß∞˚Ω·πππ€≤Ï£¨æßÃÂ÷–Al3+’ºæ›O2-–Œ≥…µƒ∞À√ÊÃÂø’œ∂£ª∏√æß∞˚µƒÃª˝Œ™(2a°¡10-7)3£¨∏√æß∞˚µƒ÷ ¡øŒ™32°¡16+16°¡27+8°¡59NA=8(59+2°¡27+4°¡16)NA£¨À˘“‘√‹∂»Œ™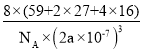 £ª
£ª
π ¥∞∏Œ™£∫∞À√ÊÃÂø’œ∂£ª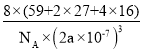 °£
°£

°æƒø°ø∏fl¬Ø¡∂Ã˙ «œ÷¥˙∏÷Ã˙…˙≤˙µƒ÷ÿ“™ª∑Ω⁄£¨¥À∑®π§“’ºÚµ•£¨≤˙¡ø¥Û£¨ƒ‹∫ƒµÕ£¨»‘ «œ÷¥˙¡∂Ã˙µƒ÷˜“™∑Ω∑®£¨ªÿ¥œ¬¡–Œ £∫
(1)“—÷™¡∂Ã˙π˝≥õƒ÷˜“™∑¥”¶Œ™Fe2O3(s)+3CO(g)=2Fe(s)+3CO2(g) °˜H1
ªπª·∑¢…˙3Fe2O3(s)+CO(g)=2Fe3O4(s)+CO2(g) °˜H2=a kJ/mol£ª
Fe3O4(s)+CO(g)=3FeO(s)+CO2(g) °˜H3=b kJ/mol£ª
FeO(s)+CO(g)=Fe(s)+ CO2(g) °˜H4=c kJ/mol£ª
‘Ú°˜H1=________kJ/mol£®”√∫¨a°¢b°¢cµƒ¥˙ ˝ Ω±Ì æ£©°£
(2)∏fl¬Ø¡∂Ã˙≤˙…˙µƒ∑œ∆¯(CO°¢CO2)”–∂‡÷÷¥¶¿Ì∑Ω∑®£¨∑¥”¶‘≠¿Ì»Áœ¬£∫
¢Ÿ…˙≥…º◊¥º£∫CO(g)+2H2(g)![]() CH3OH(l)°£“ª∂®Œ¬∂»œ¬‘⁄∫„»›√‹±’»›∆˜÷–ƒ£ƒ‚¥À∑¥”¶£¨œ¬¡–ªØ—ß∑¥”¶ÀŸ¬ ◊Óøϵƒ «_______________
CH3OH(l)°£“ª∂®Œ¬∂»œ¬‘⁄∫„»›√‹±’»›∆˜÷–ƒ£ƒ‚¥À∑¥”¶£¨œ¬¡–ªØ—ß∑¥”¶ÀŸ¬ ◊Óøϵƒ «_______________
A£Æ¶‘(CO)=1.2 mol/(L°§min) B£Æ¶‘(H2)=0.025 mol/(L°§s) C£Æ¶‘(CH3OH)=1 mol/(L°§min)
¢⁄…˙≥…““œ©£∫2CO2(g)+6H2(g)![]() C2H4(g)+4H2O(g)°£‘⁄¡Ω∏ˆ»›ª˝œ‡Õ¨µƒ√‹±’»›∆˜÷–“‘≤ªÕ¨µƒ«‚ú±»[n(H2)£∫n(CO2)]≥‰»Î‘≠¡œ∆¯£¨CO2∆Ω∫‚◊™ªØ¬ ¶¡(CO2)”ÎŒ¬∂»µƒπÿœµ»ÁÕºÀ˘ æ
C2H4(g)+4H2O(g)°£‘⁄¡Ω∏ˆ»›ª˝œ‡Õ¨µƒ√‹±’»›∆˜÷–“‘≤ªÕ¨µƒ«‚ú±»[n(H2)£∫n(CO2)]≥‰»Î‘≠¡œ∆¯£¨CO2∆Ω∫‚◊™ªØ¬ ¶¡(CO2)”ÎŒ¬∂»µƒπÿœµ»ÁÕºÀ˘ æ
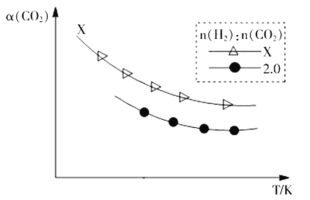
«‚ú±»X_____2.0£®Ãӣ氢£ºªÚ£Ω£¨œ¬Õ¨£©£¨¿Ì”… «___________________°£
(3)∑¥”¶CO2(g)+3H2(g)![]() CH3OH(g)+H2O(g)£¨∆‰ªØ—ß∆Ω∫‚≥£ ˝K”ÎŒ¬∂»Tπÿœµ»Á±Ì£∫
CH3OH(g)+H2O(g)£¨∆‰ªØ—ß∆Ω∫‚≥£ ˝K”ÎŒ¬∂»Tπÿœµ»Á±Ì£∫
T/°Ê | 700 | 800 | 1000 | 1200 |
K | 0.6 | 1.0 | 2.3 | 3.6 |
¢Ÿ∏√∑¥”¶ «___________∑¥”¶(ÃÓ°∞Œ¸»»°±ªÚ°∞∑≈»»°±)°£
¢⁄»Ùƒ≥∫„∂®Œ¬∂»œ¬£¨œÚ»›ª˝Œ™1 Lµƒ∫„»›√‹±’»›∆˜÷–≥‰»Î2 mol CO2°¢3 mol H2£¨10 min∫Û∑¥”¶¥ÔµΩ∆Ω∫‚◊¥Ã¨£¨≤‚µ√¶‘ (CH3OH)=0.075 mol/(L°§min)£¨‘Ú¥À∑¥”¶Ãıº˛œ¬Œ¬∂»______800°Ê£®Ãӣ氢£ºªÚ£Ω£©°£
¢€»Ù∑¥”¶‘⁄1200°ÊΩ¯–– ‘—È£¨ƒ≥ ±øÃ≤‚µ√∑¥”¶»›∆˜÷–∏˜ŒÔ÷ ≈®∂»¬˙◊„πÿœµ Ω2c(CO2)°§c3(H2)=3c(CH3OH)°§c(H2O)£¨¥À ±∑¥”¶‘⁄________œÚΩ¯––£®ÃÓ°∞’˝°±ªÚ°∞ƒÊ°±£©°£