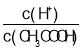��Ŀ����
����Ŀ�����ʼ�������26 ����������˰����ӵ�����(NA��6.02214076��1023 mol�D1), ������2019 �� 5��20 ����ʽ��Ч������˵������ȷ����
A. �� 1 mol![]() �У����е�������Ϊ 6 NA
�У����е�������Ϊ 6 NA
B. ��7.1 g C12����ˮ�Ƴɱ�����ˮ��ת�Ƶĵ�����Ϊ 0.1 NA
C. ��״���£�11.2 L NO��11. 2 L O2��Ϻ�����ķ�������Ϊ 0.75 NA
D. ij�¶��£�1L pH= 3�Ĵ�����Һϡ�͵�10L ʱ����Һ�� H+����Ŀ����0.01 NA
���𰸡�A
��������
A��![]() �����������11-5=6������1 mol
�к�����������11-5=6������1 mol![]() �к��е�������Ϊ 6NA����A��ȷ��
�к��е�������Ϊ 6NA����A��ȷ��
B����7.1 g C12����ˮ�Ƴɱ�����ˮ������û����ȫ��ˮ��Ӧ������ת�Ƶĵ�����С�� 0.1 NA����B����
C����״���£�11.2 L NO��11. 2 L O2��Ϻ����ڴ���2NO2![]() N2O4����������ķ�������С��0.75 NA����C����
N2O4����������ķ�������С��0.75 NA����C����
D��ij�¶��£�1L pH= 3�Ĵ�����Һϡ�͵�10L ʱ����Һ4>pH>3����Һ�� H+����ĿС��0.01 NA����D����

��ϰ��ϵ�д�
�����Ŀ