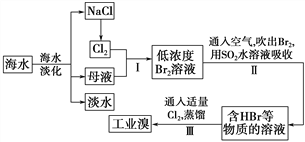
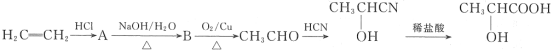��Ŀ����
����Ŀ����Ӧ4A(s)��3B(g)![]() 2C(g)��D(g)��2 min B��Ũ�ȼ���0.6 mol��L��1�������й�˵����ȷ����
2C(g)��D(g)��2 min B��Ũ�ȼ���0.6 mol��L��1�������й�˵����ȷ����
����A��ʾ�ķ�Ӧ������0.4 mol��L��1��min��1
����2 minĩʱ����Ӧ��B�ķ�Ӧ������0.3 mol��L��1��min��1
������2 min����C��ʾ��ƽ������Ϊ0.2 mol��L��1��min��1
����2 minʱ��B��C��D��Ũ�ȱ�һ��Ϊ3��2��1
����D����ʼŨ��Ϊ0.1 mol��L��1����2 minʱD��Ũ��Ϊ0.3 mol��L��1
A. �٢ڢ�B. �ڢ�C. �ܢ�D. �ۢ�
���𰸡�D
��������
��Aʹ���壬Ũ���dz����������õ�λʱ����Ũ�ȵı仯������ʾ���ٴ���
��B��2min�ڵ�ƽ����Ӧ����0.3mol/��L![]() min����2minĩ�ķ�Ӧ������˲ʱ���ʣ���0.3mol/��L
min����2minĩ�ķ�Ӧ������˲ʱ���ʣ���0.3mol/��L![]() min��С���ڴ���
min��С���ڴ���
��v(C)=![]() v��B��=0.2mol/��L
v��B��=0.2mol/��L![]() min��������ȷ��
min��������ȷ��
�ܲ�֪�����ʵ���ʼŨ�ȣ���������Ũ���뷽��ʽ�Ļ�ѧ������û�б�Ȼ��ϵ���ܴ���
�ݾ�2 min B��Ũ�ȼ���0.6 mol��L��1��B��ϵ��Ϊ3��D��ϵ��Ϊ1������������0.2 mol��L��1��D������2 minʱD��Ũ��Ϊ0.3 mol��L��1������ȷ�����Ԣۢ���ȷ��
��ѡD��

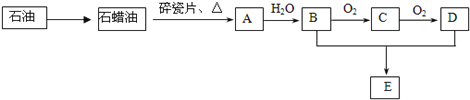
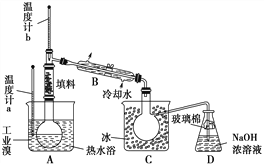
����Ŀ���ҹ��������������Ƶú�ʹ�ý���п�Ĺ��ң�һ������п��ZnS������SiO2������FeS��CdS��PbS���ʣ�Ϊԭ���Ʊ�����п��������ͼ��ʾ��

��ؽ�������[c0(Mn+)=0.1 mol��L-1]�γ��������������pH��Χ���£�
�������� | Fe3+ | Fe2+ | Zn2+ | Cd2+ |
��ʼ������pH | 1.5 | 6.3 | 6.2 | 7.4 |
������ȫ��pH | 2.8 | 8.3 | 8.2 | 9.4 |
�ش��������⣺
��1�����չ�������Ҫ��Ӧ�Ļ�ѧ����ʽΪ_______________________��
��2������1����Ҫ�ɷֳ�SiO2���___________���������ӹ�����ZnO��������____________������ͨ��������������________________��
��3����Һ�е�Cd2+����п�۳�ȥ����ԭ���ӹ����з�Ӧ�����ӷ���ʽΪ_________________��
��4���������п��Һ�Ʊ�����пʱ�������ĵ缫��ӦʽΪ______________������п��ĵ��Һ�ɷ���_______�������ʹ�á�