��Ŀ����
2����Ч����������Դ�ǽ����Դ�����һ��ʵ��;������չ��̼-��ѧ�������������ҹ���Էḻ��ú̿��Դ������Ҫ��ս������;��ü�ֵ���������Խ�̿Ϊԭ�ϣ�����̼-��ѧ��;����ȡ�Ҷ����Ĺ��̣�
��1����CO��CO2��H2Ϊԭ�ϣ���һ�������¾��ɺϳɼ״�������Ϊ�����ֺϳ���Ƹ����ϡ���ɫ��ѧ�����CO��g��+2H2��g��$\stackrel{һ������}{��}$CH3OH��g�����û�ѧ��Ӧ����ʽ��ʾ����
��2����ͼ��ʾ�ں���������230����������£�0.5 mol CO2��1.5 mol H2��Ӧ�õ��״����������ʴ�80%ʱ�������仯ʾ��ͼ��
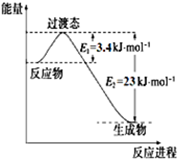
��д���÷�Ӧ���Ȼ�ѧ����ʽ��CO2��g��+3H2��g��?CH3OH��g��+H2O��g����H=-49kJ•mol-1��
�����жϸ÷�Ӧ�ﵽ��ѧƽ��״̬��������bd��������ĸ��ţ�
a��������ѹǿ���䡡����
b��H2������������䡡����
c��c��H2��=3c��CH3OH������
d���������ܶȲ���
e��2��C=O���ѵ�ͬʱ��6��H-H���ѣ�
���� ��1����CO��H2Ϊԭ�ϣ���һ�������¾��ɺϳɼ״���CO��g��+2H2��g��$\stackrel{һ������}{��}$CH3OH��g������CO2��H2Ϊԭ�ϣ���һ�������¾��ɺϳɼ״���CO2��g��+3H2��g�� $\stackrel{һ������}{��}$CH3OH��g����+H2O�����ݡ���ɫ��ѧ����˼�룬�������
��2�����ٸ���ͼ1��֪0.5mol CO2��1.5mol H2ת���ʴ�80%ʱ����23-3.4=19.6KJ��Ȼ�������㣺CO2��g��+3H2��g��?CH3OH��g��+H2O��g���ġ�H�ó��÷�Ӧ���Ȼ�ѧ����ʽ��
�ڻ�ѧƽ��ı�־�����淴Ӧ������ͬ�������Ũ�ȱ��ֲ��������ע���ѹ������
��� �⣺��1����CO��H2Ϊԭ�ϣ���һ�������¾��ɺϳɼ״���CO��g��+2H2��g��$\stackrel{һ������}{��}$CH3OH��g������CO2��H2Ϊԭ�ϣ���һ�������¾��ɺϳɼ״���CO2��g��+3H2��g�� $\stackrel{һ������}{��}$CH3OH��g����+H2O����CO��H2Ϊԭ�ϣ�����ֻ��һ���𣬷��Ϸ��ϡ���ɫ��ѧ����˼�룬�ʴ�Ϊ��CO��g��+2H2��g��$\stackrel{һ������}{��}$CH3OH��g����
��2���ٸ���ͼ1��֪0.5mol CO2��1.5mol H2ת���ʴ�80%ʱ����23-3.4=19.6KJ����÷�Ӧ���Ȼ�ѧ����ʽ��CO2��g��+3H2��g��?CH3OH��g��+H2O��g����H=-$\frac{19.6}{80%}$��2=-49kJ•mol-1���ʴ�Ϊ��CO2��g��+3H2��g��?CH3OH��g��+H2O��g����H=-49 kJ•mol-1��
��a���ں�ѹ������ѹǿʼ�ղ��䣬����֤���ﵽ��ƽ��״̬����a����
b����������� H2������������䣬˵������ֵ�������������䣬���淴Ӧ���ʣ��ﵽ��ƽ��״̬����b��ȷ��
c��c��H2��=3c��CH3OH������˵���ﵽƽ��״̬����c����
d����Ӧ���߶������壬�������������䣬��ѹ�������������ݻ��ı䣬��������ܶ��DZ�������������ܶȲ���ʱ��˵���ﵽ��ƽ��״̬����d��ȷ��
e��2��C=O���ѵ�ͬʱ��6��H-H���ѣ���ͬһ����ı仯��������״̬�¾�����������˵���ﵽƽ��״̬��
�ʴ�Ϊ��bd��
���� ���⿼���Ϊ�ۺϣ��漰��ɫ��ѧ��˼�롢�Ȼ�ѧ����ʽ����д��ƽ��״̬���жϵ�֪ʶ����Ŀ�ѶȽϴ�

| A�� | ����������NaOH��Һ��Ϻ������ã�ֱ����Ӧ��Ļ�����еμ�AgNO3��Һ������Br-�Ĵ��� | |
| B�� | ��������ϡH2SO4��Ϻ���ȣ�3min��4min��ֱ����Ӧ��Ļ�����м���������Һ�����������ǵĴ��ڣ���֪������������������ˮ��������ǣ������Ǻ�ȩ���� | |
| C�� | ���Թ��м���5mL��ˮ��2mL����0.5g���ۣ�������ã��۲��Թܵײ����ɵĺ�ɫ��״Һ�� | |
| D�� | ���Թ��м���Լ2mL������ˮ��Ȼ�����2�Ρ�3�α���ϡ��Һ���۲챽�����巴Ӧ���ɵİ�ɫ���� |
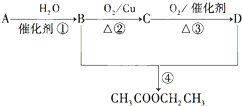
 A��B��D��E����Ԫ�ؾ�Ϊ������Ԫ�أ�ԭ������������AԪ��ԭ�ӵĺ�������������Ӳ�������������������ȣ�B��D��E����Ԫ�������ڱ��е����λ�������ʾ��ֻ��EԪ�صĵ�������ˮ��Ӧ���������ᣮ�ס��ҡ�M��W��X��Y��Z�������ʾ���A��B��D����Ԫ���е�һ�ֻ�����ɣ�����ֻ��M����ͬʱ��������Ԫ�أ�WΪA��B��Ԫ����ɵ�18���ӷ��ӣ��������ȼ�ϣ��ס���Ϊ�ǽ������ʣ�X���Ӻ���10�����ӣ�����֮���ת����ϵ��ͼ��ʾ��
A��B��D��E����Ԫ�ؾ�Ϊ������Ԫ�أ�ԭ������������AԪ��ԭ�ӵĺ�������������Ӳ�������������������ȣ�B��D��E����Ԫ�������ڱ��е����λ�������ʾ��ֻ��EԪ�صĵ�������ˮ��Ӧ���������ᣮ�ס��ҡ�M��W��X��Y��Z�������ʾ���A��B��D����Ԫ���е�һ�ֻ�����ɣ�����ֻ��M����ͬʱ��������Ԫ�أ�WΪA��B��Ԫ����ɵ�18���ӷ��ӣ��������ȼ�ϣ��ס���Ϊ�ǽ������ʣ�X���Ӻ���10�����ӣ�����֮���ת����ϵ��ͼ��ʾ��| B | D | |
| E |
��1��Z�Ļ�ѧʽΪNO2��
��2��E�ĵ�����ˮ��Ӧ�����ӷ���ʽΪCl2+H2O?H++Cl-+HClO��
��3��W����ȼ�ϵ����һ�ּ���ȼ�ϵ�أ��������Һ��20%��30%��KOH��Һ��W����ȼ�ϵ�طŵ�ʱ��������ӦʽΪO2+2H2O+4e-�T4OH-��������ӦʽΪN2H4+4OH--4e-�TN2+4H2O��
��4����һ������A2��B2�Ļ���������1L�ܱ������У���500�桢2��107Pa�´ﵽƽ�⣮���ƽ������������ʵ���Ϊ0.50mol������A2Ϊ0.3mol��B2Ϊ0.1mol�����������A2��ƽ��ת����Ϊ33.3%��
| A�� | ���뾶 | B�� | ��ѧ���� | C�� | ���������� | D�� | ������ |
| A�� | 4��3��6 | B�� | 4��3��5 | C�� | 2��5��4 | D�� | 4��6��4 |
| A�� | ˮ��ɱ�������ͣ��ܶȱ�С | |
| B�� | CH4��SiH4��GeH4��SnH4���۵�����Է���������������� | |
| C�� | He��Ne��Ar��Kr��Xe��ˮ�е��ܽ�������� | |
| D�� | ˮ���ȵ��ܸߵ��¶ȶ����Էֽ� |
 ��֪��
��֪�� ��
�� ����Ԫ�ص�ԭ������Ϊ21����Ԫ�ص�Ԫ�ط�����Sc���γɵĵ���Ϊ�������壮
����Ԫ�ص�ԭ������Ϊ21����Ԫ�ص�Ԫ�ط�����Sc���γɵĵ���Ϊ�������壮