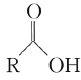
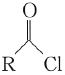
��Ŀ����
����Ŀ��a��b��c��d��ԭ��������������Ķ���������Ԫ�ء���̬bԭ��s����ϵĵ��������ǻ�̬aԭ��s����ϵĵ���������4������̬cԭ��p����ϵĵ������Ȼ�̬bԭ��p����ϵĵ�������1��b��ԭ����������d��ԭ��������һ�룬d���������������۵Ĵ�����Ϊ0������˵����ȷ���ǣ� ��
A.��̬aԭ�����������Ų�Ϊ![]()
B.����⻯������ȶ��ԣ��⣾��
C.![]() ��
��![]() ����
����![]() ������Ŀ֮��Ϊ
������Ŀ֮��Ϊ![]()
D.c����������Ԫ���γɵĶ�Ԫ�������У�c�Ļ��ϼ�һ��Ϊ-2��
���𰸡�BC
��������
a��b��c��d��ԭ��������������Ķ���������Ԫ�ء�d���������������۵Ĵ�����Ϊ0��dΪSiԪ�أ�b��ԭ����������d��ԭ��������һ�룬bΪNԪ�أ���̬cԭ��p����ϵĵ������Ȼ�̬bԭ��p����ϵĵ�������1��cΪOԪ�أ���̬bԭ��s����ϵĵ��������ǻ�̬aԭ��s����ϵĵ���������4����aΪHԪ�ء�
A��aΪHԭ�ӣ���̬aԭ�����������Ų�Ϊ![]() ����A����
����A����
B���ǽ����ԣ�N��Si������⻯������ȶ��ԣ�NH3��SiH4����B��ȷ��
C��![]() ��������
��������![]() ����
����![]() ������Ŀ֮��Ϊ
������Ŀ֮��Ϊ![]() ����C��ȷ��
����C��ȷ��
D��H2O2������-1�ۣ���D����
��ѡBC��

 ������������Ӧ����ϵ�д�
������������Ӧ����ϵ�д� ͬ����չ�Ķ�ϵ�д�
ͬ����չ�Ķ�ϵ�д�����Ŀ��̼���һˮ�ϰ�����Ҫ���������������£�����볣�����±���ʾ���ش��������⣺
������� | H2CO3 | NH3��H2O | |
���볣�� |
|
|
|
(1)̼���һ�����뷽��ʽΪ______���������볣������ʽ![]() ________��
________��
(2)Ũ�Ⱦ�Ϊ0.01 mol.L-1��H2CO3��Һ��NH3��H2O��Һ�������ϣ������Һ�е�������_______(д��ѧʽ)�������Һ��![]() ��
��![]() ��
��![]() ��
��![]() ��Ũ���ɴ�С��˳����_______��
��Ũ���ɴ�С��˳����_______��
(3)![]() ��
��![]() ��ˮ��Һ����ٽ�ˮ�⣬��ӦΪ
��ˮ��Һ����ٽ�ˮ�⣬��ӦΪ![]() �������£��÷�Ӧ��ƽ�ⳣ��
�������£��÷�Ӧ��ƽ�ⳣ��![]() _______��(����2λ��Ч����)��
_______��(����2λ��Ч����)��
(4)�����£���100 mL 0.2 mol.L-1NaHCO3��Һ�м���100 mL 0.2 mol��L-1NH3��H2O��Һ����![]() +____+_____��
+____+_____��