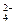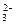��Ŀ����
ij��ɫ������ܺ���CO2��CO��H2��H2O��g���е�һ�ֻ��֣����ν������´�����ÿ����Ӧ��������ȫ������ͨ�����ȵ�̿��ָ���ԭ״̬������������䣻��ͨ�����ȵ�CuOʱ��������ɫ����ͨ����ɫCuSO4��ĩʱ����ĩ����ɫ����ͨ�����ʯ��ˮ�У�ʯ��ˮ����ǡ������ж��У���ȷ����
| A��ԭ���������һ��û��CO��H2O��һ������H2��CO2 |
| B���٢ڢۢ��ж�������������ԭ��Ӧ |
| C�����еķ�ӦΪ��CO2��3H2O��2CuSO4 ="===" Cu2��OH��2CO3��2H2SO4 |
| D���ɢ٢�˵����ʵ�������һ����CO2���� |
D
�ɢ��Ʋ�һ����CO���ɢ��Ʋ⺬H2���ɢ��Ʋ���ܺ�H2O��g������Ϊ�ڢ���Ҳ����H2O���ɢ��Ʋ���ܺ�CO2����Ϊ�ڢ���Ҳ������CO2�ڢ��й۲쵽����

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
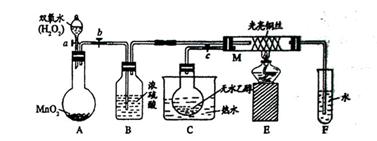
 8H2O�����NH4Cl���巴Ӧ�����е������仯�� �������������ַе���ϴ��Һ�壻 ���˷��뻥�����ܵĹ����Һ�壻 ������֪Ũ�ȵ�����ζ�����Ũ�ȵ�NaOH��Һ������к͵ζ����̣� ��ϡ��ŨH2SO4�Ĺ���
8H2O�����NH4Cl���巴Ӧ�����е������仯�� �������������ַе���ϴ��Һ�壻 ���˷��뻥�����ܵĹ����Һ�壻 ������֪Ũ�ȵ�����ζ�����Ũ�ȵ�NaOH��Һ������к͵ζ����̣� ��ϡ��ŨH2SO4�Ĺ���
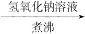 ��ȴ
��ȴ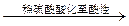
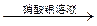 ���ְ�ɫ����
���ְ�ɫ����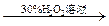
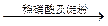 ��Һ����ɫ
��Һ����ɫ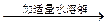
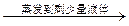
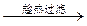
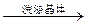 �ؽᾧ
�ؽᾧ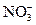 ��I-��Cl-��Fe3���е�һ�ֻ������ӣ������м�����ˮ����ˮ��ɫ�������Ʋⲻ��ȷ����
��I-��Cl-��Fe3���е�һ�ֻ������ӣ������м�����ˮ����ˮ��ɫ�������Ʋⲻ��ȷ����