��Ŀ����
��������������Ⱦ��Ϊ���أ����������������ü�����⡣
��1���������糧Ϊ��ȥ�к�����SO2�������Ϊ����������β�������գ���ͼ��ʾ��д��β���������з�Ӧ�Ļ�ѧ����ʽ�� ��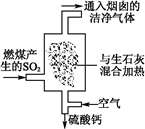
��2�������Ļ������糧ͨ�����ں��ߣ�һ�㺣ˮ�������ԣ���Ҫ����Na+��Mg2+��K+��Ca2+��Cl-��Br-��SO42-��HCO3-�����ӡ�����SO2������Ҳ�������ú�ˮ�����乤����������ͼ��ʾ��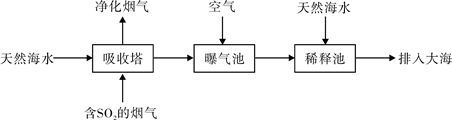
����������ͨ�������Ŀ���� ��
��ͨ���������������еĺ�ˮ����Ȼ��ˮ��ȣ�Ũ�������Բ�ͬ�������� ��
| A��Cl- | B��Na+ | C��Mg2+ | D��HCO3- |
(1) SO2��CaO=CaSO3��2CaSO3��O2=2CaSO4
��2���ٽ�H2SO3��HSO3��������ΪSO42- ��D
(3)SO2��OH��==HSO3 ��
�������������β����������Ҫ�dz�����ȼ�����ɵĶ����������SO2��CaO=CaSO3��2CaSO3��O2=2CaSO4������������������H2SO3��HSO3��������ΪSO42- ͨ���������������еĺ�ˮ����Ȼ��ˮ��ȣ�Ũ�������Բ�ͬ��������HCO3-����������̼������������ȵ�ʱ�����ֽ⡣
���㣺���黯ѧ�������м�����������֪ʶ��

 �������Ӧ���⼯ѵϵ�д�
�������Ӧ���⼯ѵϵ�д� �ۺ��Բ�ϵ�д�
�ۺ��Բ�ϵ�д���ѧ��Ԥ�⣬������ȡ�����ܣ���Ϊ��Ҫ����һ����ɫ��Դ�������й�˵������ȷ����
A��Һ����Ϊ�����Դ�ķ�Ӧԭ����4NH3��5O2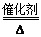 4NO��6H2O 4NO��6H2O |
| B��Һ�����и�ʴ�ԺͶ��ԣ���ʹ�ù�����Ҫ��ֹҺ��й© |
| C�������ȿ����ᣬ��״�����ܶ�ԼΪ0.76 g��L��1 |
| D��������������ȣ��ŵ����ڰ�������������ը��ʹ��ʱ����ȫ |
��ͼ��ʾ������ʢ����Һ�ٺ���Һ�ڵ��Թ��У�ͨ���������X�����բ١��ھ��г������ɵ���
| ѡ�� | X | ����Һ | ����Һ |
| A | Cl2 | Na2SO3 | Na2S |
| B | SO2 | Na2SiO3 | Ba(NO3)2 |
| C | NH3 | AgNO3 | AlCl3 |
| D | HCl | CuSO4 | NaAlO2 |
N��O��Si��S����Ҫ�ķǽ���Ԫ�أ�����˵����ȷ����
| A��N��O��S��Si��ԭ�Ӱ뾶�����ǽ��������� |
| B��������������������������γɹ⻯ѧ�����������γ��������Ҫ���� |
| C��S��SO2��Si��SiO2�������ʾ�����NaOH��Һ��Ӧ������������ijЩ�ᷴӦ |
| D��N��Si��S�ĵ��ʾ��ܺ�������Ӧ�����ɵIJ���ֱ���NO2��SiO2��SO2 |
���з�Ӧ����ʱ��������ػ�ɫ�̵��ǣ� ��
| A������������ȼ�� | B��ͭ��������ȼ�� |
| C��������������ȼ�� | D������������ȼ�� |
��10�֣���ͼ���о�ͭ��Ũ����ķ�Ӧװ�ã� 
��1��A�Թ��з�����Ӧ�Ļ�ѧ����ʽΪ ��
��2����Ӧһ��ʱ��ɹ۲쵽B�Թ��е�����Ϊ ��
��3��C�Թܿڽ���NaOH��Һ������������ ��
��4��ʵ�������֤��A�Թ��з�Ӧ���ò����Ƿ���ͭ���ӵIJ��������� ��
��5����ͭ��Ũ���ᷴӦ�Ĺ����У������к�ɫ���ʳ��֣�������������������ϡ�
| ����1 |  ����ͭ��Ũ���ᷴӦ������ɫ���ʵ�������� |
| ����2 | X���߾������������ͭ��Ũ���ᷴӦ���ɵĺ�ɫ����ΪCu2S��CuS��Cu7S4�е�һ�ֻ��֡� |
A��ͭ��Ũ���ᷴӦʱ���漰�ķ�Ӧ���ܲ�ֹһ�� B������Ũ��ѡ���ʵ����ɱ����������г��ֺ�ɫ���ʣ�C���÷�Ӧ����������֮һ������Ũ�ȡ�15 mol��L D������Ũ��Խ��ɫ����Խ����֡�Խ����ʧ





