��Ŀ����
�й�Ԫ��X��Y��Z��W����Ϣ���£�
��ش��������⣺
��1��Z��Ԫ�����ڱ��е�λ����
��2��Y��W��ȣ��ǽ����Խ�ǿ����
a��WԪ���⻯����ȶ��Դ���YԪ���⻯����ȶ���
b��WԪ���⻯��ˮ��Һ������ǿ��YԪ���⻯��ˮ��Һ������
c�� WԪ�صĵ�������YԪ�ص��⻯�ﷴӦ���û���Y����
d�� W����ۺ������Y����ۺ����������ǿ
��3��ZԪ�غ�XԪ�ؿ��γ�����ZX42-���������ӵ�������������ɫ������������������
ZX42-����ǿ�����ԣ���ɱ����������ԭ������Z3+���������ӵ���������������ԭ���ǣ������ӷ���ʽ�ͼ�Ҫ������˵����
| Ԫ�� | �й���Ϣ |
| X | ԭ�Ӱ뾶Ϊ0.074nm�����⻯��Ľṹģ�Ϳɱ�ʾΪ�� ��������Һ�� ��������Һ�� |
| Y | ԭ�Ӱ뾶Ϊ0.102nm����ԭ�Ӻ�������Ų�Ϊ��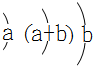 |
| Z | �����б�����Ԫ���к�����࣬����ȱʧ������ƶѪ |
| W | ��������������������������֮��Ϊ4 |
��1��Z��Ԫ�����ڱ��е�λ����
�������ڡ�����
�������ڡ�����
����2��Y��W��ȣ��ǽ����Խ�ǿ����
Cl
Cl
����Ԫ�ط��ţ���������ʵ��֤����һ���۵���acd
acd
������ĸ����a��WԪ���⻯����ȶ��Դ���YԪ���⻯����ȶ���
b��WԪ���⻯��ˮ��Һ������ǿ��YԪ���⻯��ˮ��Һ������
c�� WԪ�صĵ�������YԪ�ص��⻯�ﷴӦ���û���Y����
d�� W����ۺ������Y����ۺ����������ǿ
��3��ZԪ�غ�XԪ�ؿ��γ�����ZX42-���������ӵ�������������ɫ������������������
ZX42-����ǿ�����ԣ���ɱ����������ԭ������Z3+���������ӵ���������������ԭ���ǣ������ӷ���ʽ�ͼ�Ҫ������˵����
FeO42-�Ļ�ԭ����ΪFe3+���ܷ���ˮ�ⷴӦ��Fe3++3H2O?Fe��OH��3�����壩+3H+�����ɵ����������������������ã���ˮ����
FeO42-�Ļ�ԭ����ΪFe3+���ܷ���ˮ�ⷴӦ��Fe3++3H2O?Fe��OH��3�����壩+3H+�����ɵ����������������������ã���ˮ����
�����������⻯�ﳣ����ΪҺ̬�Լ�����ģ�Ϳ�֪XΪOԪ�أ��γɵ��⻯��Ϊˮ��
�ɺ�������Ų���֪a=2��b=6����YΪSԪ�أ�
Z�����б�����Ԫ���к�����࣬����ȱʧ������ƶѪ��ZΪFeԪ�أ�
W��������������������������֮��Ϊ4������ΪO��ClԪ�أ�����XΪOԪ�أ���WΪClԪ�أ�
���Ԫ�ض�Ӧ�ĵ��ʡ�����������ʽ����⣮
�ɺ�������Ų���֪a=2��b=6����YΪSԪ�أ�
Z�����б�����Ԫ���к�����࣬����ȱʧ������ƶѪ��ZΪFeԪ�أ�
W��������������������������֮��Ϊ4������ΪO��ClԪ�أ�����XΪOԪ�أ���WΪClԪ�أ�
���Ԫ�ض�Ӧ�ĵ��ʡ�����������ʽ����⣮
����⣺��1��ZΪFeԪ�أ�ԭ������Ϊ26��λ�����ڱ��������ڡ����壬�ʴ�Ϊ���������ڡ����壻
��2��ͬ����Ԫ�ش�����Ԫ�صķǽ���������ǿ����ǽ����Խ�ǿ��Ԫ��ΪCl���Ƚ�Ԫ�صķǽ����ԣ��ɸ���
�⻯����ȶ��ԡ���ۺ�����������Լ�����֮����û���Ӧ�������ܸ����⻯������ԣ�����������Ա�ˮǿ�����ǽ�����S�ı�O����������Ϊ�жϵ����ݣ�
�ʴ�Ϊ��Cl��acd��
��3��FeO42-����ǿ�����ԣ���Ϊ��ԭ����Fe3+��Fe3+��ˮ������Fe��OH��3���壬���н�ǿ�������ԣ�����ˮ���ã�
ˮ�ⷴӦ�����ӷ���ʽΪFe3++3H2O?Fe��OH��3�����壩+3H+��
�ʴ�Ϊ��FeO42-�Ļ�ԭ����ΪFe3+���ܷ���ˮ�ⷴӦ��Fe3++3H2O?Fe��OH��3�����壩+3H+�����ɵ����������������������ã���ˮ���ã�
��2��ͬ����Ԫ�ش�����Ԫ�صķǽ���������ǿ����ǽ����Խ�ǿ��Ԫ��ΪCl���Ƚ�Ԫ�صķǽ����ԣ��ɸ���
�⻯����ȶ��ԡ���ۺ�����������Լ�����֮����û���Ӧ�������ܸ����⻯������ԣ�����������Ա�ˮǿ�����ǽ�����S�ı�O����������Ϊ�жϵ����ݣ�
�ʴ�Ϊ��Cl��acd��
��3��FeO42-����ǿ�����ԣ���Ϊ��ԭ����Fe3+��Fe3+��ˮ������Fe��OH��3���壬���н�ǿ�������ԣ�����ˮ���ã�
ˮ�ⷴӦ�����ӷ���ʽΪFe3++3H2O?Fe��OH��3�����壩+3H+��
�ʴ�Ϊ��FeO42-�Ļ�ԭ����ΪFe3+���ܷ���ˮ�ⷴӦ��Fe3++3H2O?Fe��OH��3�����壩+3H+�����ɵ����������������������ã���ˮ���ã�
���������⿼��Ԫ�صĽṹ��Ԫ��������֪ʶ����Ŀ�ѶȲ�����ע�����ԭ�Ӻ�������Ų��ص㣬Ϊ������Ĺؼ���ѧϰ��ע����ձȽϷǽ����Ե�ǿ���ķ�����

��ϰ��ϵ�д�
�����Ŀ


 �й�Ԫ��X��Y��Z��D��E����Ϣ����
�й�Ԫ��X��Y��Z��D��E����Ϣ����