��Ŀ����
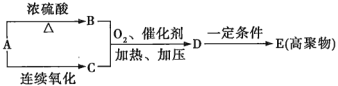
����Ŀ��ȼú�������г����д�����NOx��CO��SO2���к����壬������Ⱦ�������к�����Ի�����Ӱ���ǻ�ѧ�������о�����Ҫ���⡣
��.��֪��������ɲ���ǿ������Na2S2O8�����ѳ�������������ʾ��

(1)д����Ӧ1�����ӷ���ʽ��____ ��
(2)��Ӧ2ΪNO2-��S2O82-��2OH��=NO3-��2SO42-��H2O���˷�Ӧ�ڲ�ͬ�¶��´ﵽƽ��ʱ��NO2-���ѳ�����Na2S2O8��ʼŨ�ȵĹ�ϵ��ͼ1��ʾ��

�ٱȽ�a��b��ķ�Ӧ���ʣ�va��____vb��(����>����<����������)��
�ڸ���ͼ������жϸ÷�ӦΪ���ȷ�Ӧ��������____________��
��.�������������Ϊ��Ӧ2CO(g)��O2(g)![]() 2CO2(g)�Ĵ�����ͼ2��ʾ����ͬ�ĺ����ܱ���������ͬ����ʼŨ�ȡ���ͬ�ķ�Ӧʱ����£�ʹ����ͬ�����IJ�ͬ���������(���͡�����)��ʱ��CO��ת�������¶ȵĹ�ϵ��
2CO2(g)�Ĵ�����ͼ2��ʾ����ͬ�ĺ����ܱ���������ͬ����ʼŨ�ȡ���ͬ�ķ�Ӧʱ����£�ʹ����ͬ�����IJ�ͬ���������(���͡�����)��ʱ��CO��ת�������¶ȵĹ�ϵ��

(3)���й���ͼ2��˵����ȷ����________��
A���ھ�δ�ﵽƽ��״̬ʱ��ͬ�������������������COת�����ʱ�����Ҫ��
B��b��ʱCO��O2����֮�䷢����Ч��ײ�ļ���������ʵ����������
C��a��b��c��d�ĵ��У��ﵽƽ��״̬��ֻ��b��c����
D��e��ת���ʳ���ͻ���ԭ��������¶����ߺ����ʧȥ����
(4)��֪c��ʱ������O2Ũ��Ϊ0.02 mol/L����50 ��ʱ�������������������COת����Ӧ��ƽ�ⳣ��K��_____________(�ú�x�Ĵ���ʽ��ʾ)��
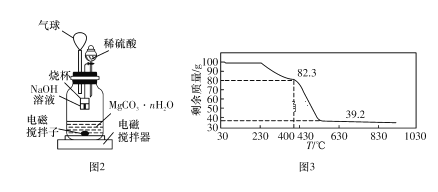
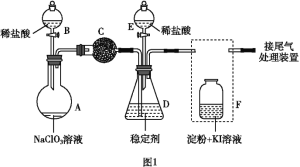
��.����ͼ3��ʾװ��(�缫��Ϊ���Ե缫)������SO2��

(5)ֱ����Դb��Ϊ____�����������ķ�ӦΪ______��
���𰸡�2NO��S2O82-��4OH��===2NO2-��2SO42-��2H2O < ��Na2S2O8��ʼŨ����ͬʱ���¶�Խ�ߣ�NO![]() �ѳ���Խ��˵��ƽ�������ƶ����÷�ӦΪ���ȷ�Ӧ AD
�ѳ���Խ��˵��ƽ�������ƶ����÷�ӦΪ���ȷ�Ӧ AD ![]() mol/L ���� SO2��2e����2H2O===SO42-��4H��
mol/L ���� SO2��2e����2H2O===SO42-��4H��
��������
���������֪�����⿼�黯ѧƽ���Ӱ�����ؼ���ѧƽ��ļ��㣬������������ԭ�����з�����
��1����Ӧ1��Na2S2O8������������Һ��ͨ��NO���巢��������ԭ��Ӧ���������ơ��������ƺ�ˮ, ��Ӧ�����ӷ���ʽΪ��2NO��S2O82-��4OH��===2NO2-��2SO42-��2H2O,
�ʴ�Ϊ��2NO��S2O82-��4OH��===2NO2-��2SO42-��2H2O��
��2���¶�Խ��NO�ѳ���Խ��˵�����·�Ӧ��������a��b��ķ�Ӧ����va��< vb����
�ʴ�Ϊ��<��
��3������ƽ�������ȷ����ƶ�������Na2S2O8��ʼŨ����ͬʱ���¶�Խ�ߣ�NO![]() �ѳ���Խ��˵��ƽ��������У�����ӦΪ���ȷ�Ӧ��
�ѳ���Խ��˵��ƽ��������У�����ӦΪ���ȷ�Ӧ��
�ʴ�Ϊ����Na2S2O8��ʼŨ����ͬʱ���¶�Խ�ߣ�NO![]() �ѳ���Խ��˵��ƽ�������ƶ����÷�ӦΪ���ȣ�
�ѳ���Խ��˵��ƽ�������ƶ����÷�ӦΪ���ȣ�
(3)A. ��ͼ�ã���ͬ�¶��¦��������������CO�������ʱ�����Ҫ��A��ȷ��
B. a������ݷ�ӳ��CO������������ʵ���������ߵģ���B����
C. ͼ�������֪b�㷴Ӧ�ﵽƽ��״̬������ƽ��������У�a��b��c��d �ĵ��У��ﵽƽ��״̬����bcd, ��C����
D. e�㽵���ʳ���ͻ���ԭ��������¶����ߺ����ʧȥ����ʧЧ����D��ȷ��
�ʴ�Ϊ��AD��
��4������ʼʱCO��Ũ��Ϊ1mol��L��1����ﵽƽ��ʱc(CO)=(1��x)mol/L��c(O2)=0.02mol/L��c(CO2)=xmol/L������ƽ�ⳣ���Ķ��壬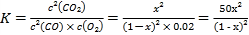
�ʴ�Ϊ��![]() ��
��
��5����װ��ͼ��֪SO2����߷���������Ӧ����SO42-���缫��Ӧ����ʽΪ��SO2��2e����2H2O===SO42-��4H�������a�����ĵ缫Ϊ���ص������������ص��������ӵ��ǵ�Դ�����������aΪ��Դ������bΪ��Դ������
�ʴ�Ϊ��������SO2��2e����2H2O===SO42-��4H����

����Ŀ�������ȣ�ClNO�����л��ϳ��е���Ҫ�Լ����������ȿ���NO��Cl2��ͨ�������·�Ӧ�õ�,��ѧ����ʽΪ2NO��g��+Cl2��g�� ![]() 2ClNO��g����
2ClNO��g����
��1�����������������ڴ����еĺ������������ʱ��������������,�漰���·�Ӧ:
��4NO2��g��+2NaCl��s�� ![]() 2NaNO3��s��+2NO��g��+Cl2��g�� K1
2NaNO3��s��+2NO��g��+Cl2��g�� K1
��2NO2��g��+NaCl��s��![]() NaNO3��s��+ClNO��g�� K2
NaNO3��s��+ClNO��g�� K2
��2NO��g��+Cl2��g��![]() 2ClNO��g�� K3
2ClNO��g�� K3
��K1,K2,K3֮��Ĺ�ϵΪK3=______________��
��2��T��ʱ,2NO��g��+Cl2��g�� ![]() 2ClNO��g��������Ӧ���ʱ���ʽΪv��= k cn��ClNO��,������ʺ�Ũ�ȵĹ�ϵ���±�:
2ClNO��g��������Ӧ���ʱ���ʽΪv��= k cn��ClNO��,������ʺ�Ũ�ȵĹ�ϵ���±�:
��� | c��ClNO��/mol��L-1 | v/mol��L-1��s-1 |
�� | 0.30 | 3.6��10-8 |
�� | 0.60 | 1.44��10-7 |
�� | 0.90 | 3.24��10-7 |
n=____________;k=____________��ע����λ����
��3����2L�ĺ����ܱ������г���4mol NO��g����2mol Cl2��g��,�ڲ�ͬ�¶��²��c��ClNO����ʱ��Ĺ�ϵ��ͼA
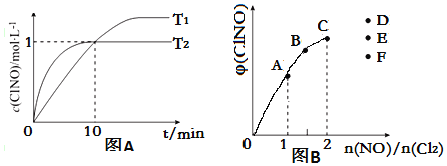
���¶�ΪT1ʱ,����Ϊ�÷�Ӧ�ﵽƽ��ı�־����__________;
a ����������ֲ���
b ����ѹǿ���ֲ���
c ƽ�ⳣ��K���ֲ���
d ������ɫ���ֲ���
e v��ClNO��= v��NO��
f NO��ClNO�����ʵ�����ֵ���ֲ���
�ڷ�Ӧ��ʼ��10minʱCl2��ƽ����Ӧ����v��Cl2��=____________;
���¶�ΪT2ʱ,10min�Ѿ��ﵽƽ��,�÷�Ӧ��ƽ�ⳣ��K
��4��һ���������ں��º��ݵ��ܱ������а�һ����������NO��g����Cl2��g��,ƽ��ʱClNO�������������n��NO��/n��Cl2���ı仯ͼ����ͼB,��A��B��C��״̬��,NO��ת������С����__________��,��n��NO��/n��Cl2��=2.8ʱ,�ﵽƽ��״̬ClNO�����������������D��E��F�����е�__________�㡣
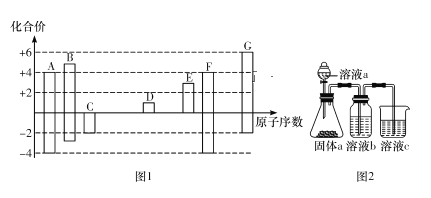
����Ŀ��Ԫ��A��D��Ԫ�����ڱ��ж����ڵ�����Ԫ�أ�����ݱ�����Ϣ�ش��������⡣
Ԫ�� | A | B | C | D |
���ʻ�ṹ��Ϣ | �����Ƴɵĸ�ѹ�Ʒ����Ļƹ�����ǿ�����Զ | ��ҵ��ͨ������Һ̬��������䵥�ʡ�ԭ�ӵ������δ�ﵽ�ȶ��ṹ | �����ڳ��¡���ѹ��������,ԭ�ӵ� | 2�������ӵĺ�������Ų�����ԭ����ͬ |
��1��A��Cl�γɵĻ�����Ϊ__________������ӡ����ۡ������������__________����
��2��D��C�γɵĻ������к���__________����д��C������ˮ��Ӧ�Ļ�ѧ����ʽ��____________________��
��3�����ж�Ԫ��B��Ԫ��B�ij�������������ȷ����__________������ţ���
a.BԪ�ص��������Ϊ+6
b.���¡���ѹ��B�ij�������������ˮ
c.B�ij������ʷ����к���18������
d.��һ��������þ������B�ij������ʷ�Ӧ
��4����B��H���γ�![]() ����BΪ__________Ԫ�أ�
����BΪ__________Ԫ�أ�![]() �к��еĻ�ѧ��Ϊ__________��
�к��еĻ�ѧ��Ϊ__________��
��5��A��D��Ԫ���н����Խ�ǿ����__________��дԪ�ط��ţ���д����֤���ý��۵�һ��ʵ����ʵ��____________________��