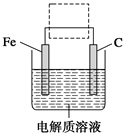
��Ŀ����
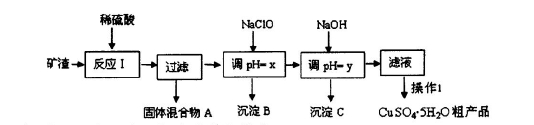
ij�����ijɷ�ΪCu2O��Al2O3��Fe2O3��SiO2����ҵ���øÿ�����ȡͭ�͵����IJ����������£�
��֪�� ��Cu2O +2 H+="Cu" + Cu2++H2O
�ڲ���������������������ʽ����ʱ��Һ��pH���±���
��ش��������⣺
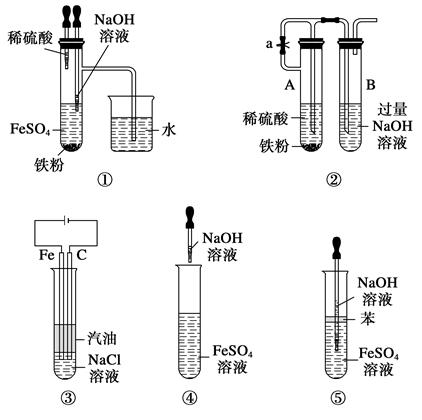
��1��Ϊ�˼ӿ췴ӦI�����ʣ����Բ�ȡ�Ĵ�ʩ�� (д2��)��
��2����������A�еijɷ��� ��
��3����ӦI��ɺ���Ԫ�صĴ�����ʽΪ (�����ӷ���)��д�����ɸ����ӵ����ӷ���ʽ ��
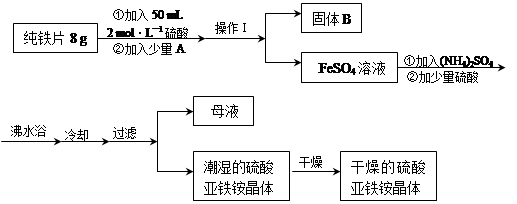
��4������1��Ҫ������ �� �� ��ϴ��CuSO4?5H2O�ֲ�Ʒ�����ô���ˮϴ�����ñ�ˮϴ�ӡ�ԭ���� ��
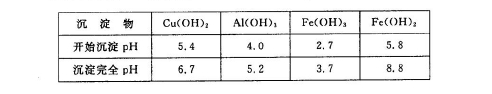
��5���ö��Ե缫������Һһ��ʱ�䣬����0��1 mol��Cu(OH)2�ɻָ���Һԭ��(Ũ�ȡ��ɷ�)������ʱת�Ƶ��ӵ����ʵ���Ϊ ����
��6����NaClO��pH�����ɳ���B��ͬʱ����һ�־���Ư�����õ����ʣ��÷�Ӧ�����ӷ���ʽΪ ��

��֪�� ��Cu2O +2 H+="Cu" + Cu2++H2O
�ڲ���������������������ʽ����ʱ��Һ��pH���±���

��ش��������⣺
��1��Ϊ�˼ӿ췴ӦI�����ʣ����Բ�ȡ�Ĵ�ʩ�� (д2��)��
��2����������A�еijɷ��� ��
��3����ӦI��ɺ���Ԫ�صĴ�����ʽΪ (�����ӷ���)��д�����ɸ����ӵ����ӷ���ʽ ��
��4������1��Ҫ������ �� �� ��ϴ��CuSO4?5H2O�ֲ�Ʒ�����ô���ˮϴ�����ñ�ˮϴ�ӡ�ԭ���� ��
��5���ö��Ե缫������Һһ��ʱ�䣬����0��1 mol��Cu(OH)2�ɻָ���Һԭ��(Ũ�ȡ��ɷ�)������ʱת�Ƶ��ӵ����ʵ���Ϊ ����
��6����NaClO��pH�����ɳ���B��ͬʱ����һ�־���Ư�����õ����ʣ��÷�Ӧ�����ӷ���ʽΪ ��
��1���ʵ������¶ȣ����Ͻ��裻���������飻�ʵ����������Ũ�ȵȣ�
��2��SiO2��Cu
��3��Fe3+ 2Fe3++Cu=Cu2++2Fe2+
��4������Ũ������ȴ�ᾧ�����ˣ���ˮ�ȿ���ϴȥ���������������ӣ��ֿ��Լ��پ������ʧ��
��5��0��4mol
��6��2Fe2++7ClO-+2H++5H2O=2Fe(OH)3+Cl-+6HClO
��2��SiO2��Cu
��3��Fe3+ 2Fe3++Cu=Cu2++2Fe2+
��4������Ũ������ȴ�ᾧ�����ˣ���ˮ�ȿ���ϴȥ���������������ӣ��ֿ��Լ��پ������ʧ��
��5��0��4mol
��6��2Fe2++7ClO-+2H++5H2O=2Fe(OH)3+Cl-+6HClO
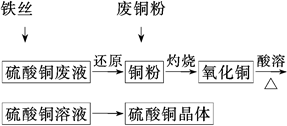
�����������1�����ڿ���Ϊ���壬����Ϊ�˼ӿ췴ӦI�����ʣ����Բ�ȡ�Ĵ�ʩ�ǽ�������顣Ҳ�����ʵ����߷�Ӧ���¶ȣ�������Ӧ��H2SO4��Ũ�ȵȴ�ʩ����2��SiO2������������������ᷢ����Ӧ����������������Cu2O��Al2O3��Fe2O3�����ᷢ����Ӧ��Cu2O+H2SO4=Cu+CuSO4+H2O��Al2O3+3H2SO4= Al2(SO4)3+3H2O��Fe2O3+3H2SO4= Fe2(SO4)3+3H2O�� Fe2(SO4)3+Cu= CuSO4+ 2FeSO4����˷�����ӦI����˳��Ĺ�������A�еijɷ���SiO2��Cu����3����Ϊ�ڿ�����FeԪ�صĻ��ϼ�Ϊ+3�ۣ��ȷ������ֽⷴӦ���õ�Fe2(SO4)3,Ȼ������Ӧ��2Fe3++Cu=Cu2++2Fe2+����˸���ɺ���Ԫ�صĴ�����ʽΪ+2�۵�Fe����Fe2+����4��������ͭ��Һ����ȡ����ͭ����IJ���1��Ҫ������ ����Ũ������ȴ�ᾧ�����ˡ�ϴ��CuSO4?5H2O�ֲ�Ʒ�����ô���ˮϴ�����ñ�ˮϴ�ӡ�ԭ����CuSO4��Һ�ܽ���ˮ�������¶�Խ�ߣ��ܽ��Խ�����ñ�ˮ�ȿ���ϴȥ���������������ӣ��ֿ��Լ��پ������ʧ����5���ö��Ե缫������Һ��ⷴӦ����ʽΪ��2CuSO4+2H2O
 2Cu+O2��+2H2SO4��������0��1 mol��Cu(OH)2�ᷢ����Ӧ��Cu(OH)2+H2SO4= CuSO4+2H2O ������0��1mol��CuSO4��0��2mol��ˮ�ָ�ԭ������Һ������ݵ�ⷽ��ʽ�е����ʼ�Ĺ�ϵ��֪����ת��0��4mol����6��NaClO�������ԣ�Fe2+�л�ԭ�ԡ�������NaClO����pH�����ɳ���BΪFe(OH)3��ͬʱ����һ�־���Ư�����õ�����ΪHClO���÷�Ӧ�����ӷ���ʽΪ2Fe2++7ClO-+2H++5H2O=2Fe(OH)3��+Cl-+6HClO
2Cu+O2��+2H2SO4��������0��1 mol��Cu(OH)2�ᷢ����Ӧ��Cu(OH)2+H2SO4= CuSO4+2H2O ������0��1mol��CuSO4��0��2mol��ˮ�ָ�ԭ������Һ������ݵ�ⷽ��ʽ�е����ʼ�Ĺ�ϵ��֪����ת��0��4mol����6��NaClO�������ԣ�Fe2+�л�ԭ�ԡ�������NaClO����pH�����ɳ���BΪFe(OH)3��ͬʱ����һ�־���Ư�����õ�����ΪHClO���÷�Ӧ�����ӷ���ʽΪ2Fe2++7ClO-+2H++5H2O=2Fe(OH)3��+Cl-+6HClO
��ϰ��ϵ�д�
�����Ŀ
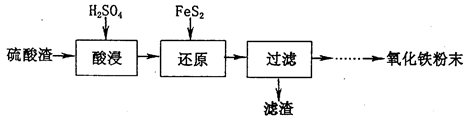
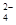 ���÷�Ӧ�����ӷ���ʽΪ____________.
���÷�Ӧ�����ӷ���ʽΪ____________.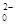
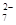 +14H+===6Fe3++2Cr3++7H2O
+14H+===6Fe3++2Cr3++7H2O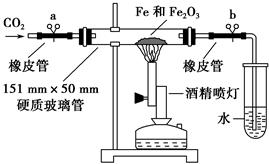
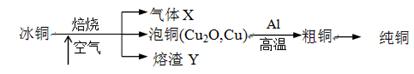

 6SO2��Fe3O4����3 mol FeS2�μӷ�Ӧ��ת��________ mol���ӡ�
6SO2��Fe3O4����3 mol FeS2�μӷ�Ӧ��ת��________ mol���ӡ�