��Ŀ����
����Ŀ���ı�����ϩ(TPE)��������������յ��������ԣ��ڹ����ϵ�����Ӧ��ǰ���㷺��������TPE�������ϳ�·��(�����Լ�����Ӧ����ʡ��)��

��1��A��������__________________���Լ�YΪ____________________��
��2��B��C�ķ�Ӧ����Ϊ_______________��B�й����ŵ�������_________________��D�й����ŵ�������_______________��
��3��E��F�Ļ�ѧ����ʽ��______________________________________��
��4��W��D��ͬ���칹�壬�������нṹ����������������![]() ����һԪȡ����ڴ����Ǽ�(��CH2OH)��д��W���п��ܵĽṹ��ʽ��______________________��
����һԪȡ����ڴ����Ǽ�(��CH2OH)��д��W���п��ܵĽṹ��ʽ��______________________��
��5������������ȷ����______��
a. B�����Աȱ���ǿ
b. D���ܷ�����ԭ��Ӧ
c. E����3�ֲ�ͬ��ѧ��������
d. TPE�����ڷ�����Ҳ����ϩ��
���𰸡��ױ� ���Ը������ ȡ����Ӧ �Ȼ� �ʻ� 
![]() ��
�� a d
a d
��������
A�ķ���ʽΪC7H8��A��ת��Ϊ ����A�Ľṹ��ʽΪ
����A�Ľṹ��ʽΪ![]() ������E��F��E�Ľṹ��ʽ��F�ķ���ʽ��E����ˮ�ⷴӦ����F��F�Ľṹ��ʽΪ
������E��F��E�Ľṹ��ʽ��F�ķ���ʽ��E����ˮ�ⷴӦ����F��F�Ľṹ��ʽΪ ��
��
��1��A�Ľṹ��ʽΪ![]() ��A������Ϊ�ױ����ױ�����������Ҫ���Լ�YΪ���Ը�����أ�
��A������Ϊ�ױ����ױ�����������Ҫ���Լ�YΪ���Ը�����أ�
��2���Ա�B��C�Ľṹ��ʽ��B��CΪȡ����Ӧ��B�Ľṹ��ʽΪ ��B�й����ŵ�����Ϊ�Ȼ���D�Ľṹ��ʽΪ
��B�й����ŵ�����Ϊ�Ȼ���D�Ľṹ��ʽΪ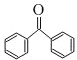 ��D�й����ŵ�����Ϊ�ʻ���
��D�й����ŵ�����Ϊ�ʻ���
��3��E��F�Ļ�ѧ����ʽ�� ��
��
��4��D�Ľṹ��ʽΪ ��D�ķ���ʽΪC13H10O�������Ͷ�Ϊ9��D��ͬ���칹��W���Т���������
��D�ķ���ʽΪC13H10O�������Ͷ�Ϊ9��D��ͬ���칹��W���Т���������![]() ����һԪȡ���������7�������Ͷȣ����������2�������Ͷȣ��ڴ����Ǽ�(��CH2OH)������������W�Ľṹ��ʽΪ
����һԪȡ���������7�������Ͷȣ����������2�������Ͷȣ��ڴ����Ǽ�(��CH2OH)������������W�Ľṹ��ʽΪ![]() ��
�� ��
��
��5��a. B�Ľṹ��ʽΪ ���Ȼ������Դ��ڷ��ǻ���B�����Աȱ���ǿ����a��ȷ��
���Ȼ������Դ��ڷ��ǻ���B�����Աȱ���ǿ����a��ȷ��
b. D�Ľṹ��ʽΪ ��D���������������ӳɷ�Ӧ������ԭ��Ӧ����b����
��D���������������ӳɷ�Ӧ������ԭ��Ӧ����b����
c.E�Ľṹ��ʽΪ ��E�к���4�ֲ�ͬ��������ԭ�ӣ���c����
��E�к���4�ֲ�ͬ��������ԭ�ӣ���c����
d.TPE�����к��б�����̼̼˫����TPE�����ڷ�����Ҳ����ϩ������d��ȷ����ѡad��

 ��У����ϵ�д�
��У����ϵ�д�����Ŀ��ij�о�С��������Ϸ���Fe3+��I-�ķ�Ӧ���п����ԣ��Ʋ��������뱥�͵�ˮ�Ļ��Һ�л����Fe3+�����ݴ����ʵ��1������ͼ��ʾ����������Һδ��졣��С�����Һδ����ԭ�����������̽����
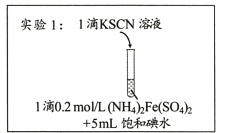
I������̽��
ʵ��2���������ȴ�������ˮ�����������ֱ�����Һ����ʵ�飬ʵ���¼���±���
��� | �� �� ��NH4��2Fe��SO4��2��Һ | ���͵�ˮ | ʯ���� | ���� | KSCN��Һ | ���� |
2-1 | 1�� | 5mL | 1mL | ������ | 1�� | ���������� |
2-2 | 1�� | 5mL | 1mL | ����һ��ʱ�����ȴ | 1�� | ���������� |
(1)�о�С��ͬѧ��������������д����Fe3+��I-��Ӧ�����ӷ���ʽΪ____��
(2)ʵ��2�У�����ʯ���͵�Ŀ����____��
(3)С��ͬѧ�����ƶϣ���Ӧ���ʲ��ǵ���ʵ��1����Һδ������Ҫԭ�����ǵ�������___��
II���������ϣ�����̽��
�����ϣ�AgSCNΪ������ˮ�İ�ɫ���塣
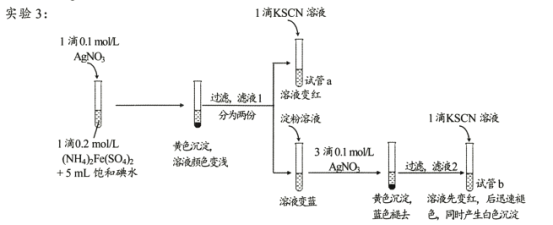
(4)�Թ�a��ʵ������˵����Һl�к���____��
(5)�Թ�b�У�����KSCN��Һ��������ʵ�������ԭ���� ___��
(6)��ͬѧ��ʵ��3��������ɣ���Ϊ��ʵ��������֤��I2������Fe2+����������¼��裺i��������AgNO3������Fe2+;ii�������ǿ����е�O2������Fe2+�� ����ơ�ʵʩ��ʵ��4��ʵ���¼���±���
��� | ʵ����� | ʵ������ |
4 | ���Թ��м���1��0.2mol/L��NH4��2Fe��SO4��2��Һ��5mL����ˮ��1���Լ�a��һ��ʱ������1��KSCN��Һ | ��Һ����� |
���Լ�a�� ___��
������ʵ��4����ͬѧ�����жϣ�______________________
(7)����ʵ��1��4���ý�����____��
����Ŀ��Li4Ti5O12��LiFePO4��������ӵ�صĵ缫���ϣ���������������Ҫ�ɷ�ΪFeTiO3������������MgO��SiO2�����ʣ����Ʊ��������������£�

�ش��������⣺
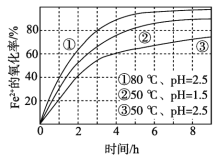
��1���������ʵ���У����Ľ����ʽ������ͼ��ʾ����ͼ��֪�������ľ�����Ϊ70%ʱ�������õ�ʵ������Ϊ___________________��

��2���������������Ҫ��TiOCl42����ʽ���ڣ�д����Ӧ��Ӧ�����ӷ���ʽ__________________��
��3��TiO2��xH2O������˫��ˮ����ˮ��Ӧ40 min����ʵ�������±���ʾ��
�¶�/�� | 30 | 35 | 40 | 45 | 50 |
TiO2��xH2Oת����% | 92 | 95 | 97 | 93 | 88 |
����40��ʱTiO2��xH2Oת������ߵ�ԭ��__________________��
��4��Li2Ti5O15��Ti�Ļ��ϼ�Ϊ+4�����й���������ĿΪ__________________��
��5��������Һ�ڡ���c(Mg2+)=0.02 mol/L������˫��ˮ�����ᣨ����Һ�������1������ʹFe3+ǡ�ó�����ȫ����Һ��c(Fe3+)=1��10-5 mol/L����ʱ�Ƿ���Mg3(PO4)2�������ɣ�___________����ʽ���㣩��
FePO4��Mg3(PO4)2��Ksp�ֱ�Ϊ1.3��10-22��1.0��10-24��
��6��д�����������բڡ�����FePO4�Ʊ�LiFePO4�Ļ�ѧ����ʽ______��