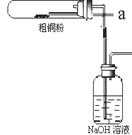
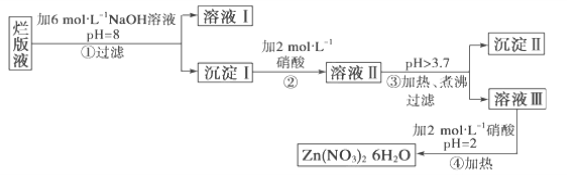
��Ŀ����
����Ŀ��ijѧ�����к͵ζ����ⶨij�ռ���Ʒ�Ĵ��ȡ���ȷ����10.0g�������������������ʵ���Ʒ�����500mL������Һ����0.200mol/L�������Һ�ζ�����Һ���ζ�ʱ�÷�̪��ָʾ�����Ը���ʵ��ش��������⣺
��1�������йصζ�������˳����ȷ����
���ñ���Һ��ϴ�ζ��� �����ζ�����ע�����Һ
�����ζ����Ƿ�©ˮ ���ζ� ��ϴ��
A���ݢ٢ڢۢ� B���ۢݢ٢ڢ� C���ۢݢڢ٢� D���ڢ٢ۢݢ�
��2���ñ�������ζ������NaOH��Һʱ��һ������ʽ�ζ��ܵĻ�����һ��ҡ����ƿ���۾�ע��_________����ʵ��ζ��ﵽ�յ�ı�־��_________________________��
��3�������±����ݣ����㱻���ռ���Һ�����ʵ���Ũ���� ���ռ���Ʒ�Ĵ�����___________
�ζ����� | ������Һ���(mL) | ������� | |
�ζ�ǰ�̶�(mL) | �ζ���̶�(mL) | ||
��һ�� | 10.00 | 0.40 | 20.50 |
�ڶ��� | 10.00 | 4.10 | 24.00 |
������ | 10.00 | 0.20 | 24.20 |
��4������ʵ���ijɡ��ô����Һ�ζ������Һ���������в�����ʹ�ռ���Ʒ����ƫ�ߵ���
A����ƿ������ˮϴ�������������Һ��ϴ��װ��Һ��
B����ʽ�ζ���������ˮϴ����δ��ϴ
C���ζ�ǰ��ʽ�ζ��ܼ������δ�ų����ζ���������ʧ
D���ζ�ǰ���Ӽ�ʽ�ζ��ܶ������ζ����ӵζ��ܶ���
���𰸡�
��1)B
��2����ƿ����Һ����ɫ�仯 ��Һ��ɫ�ɺ�ɫͻ�����ɫ���Ұ�����ڲ��ָ�ԭ������ɫ��
��3��0.400 80% ��4��D
��������
�����������1���к͵ζ��У����ռ�©��ϴ�ӡ���ϴ��װҺ���ζ��Ȳ������У�����ȷ�ĵζ�˳��Ϊ���ۢݢ٢ڢ�����ѡB��
��2������к͵ζ�ʱ���۾�Ҫע����ƿ����Һ����ɫ�仯���ζ��յ�ʱ��Һ��ɫ�ɺ�ɫͻ��Ϊ��ɫ���Ұ�����ڲ��ָ�ԭ������ɫ��
��3)�������ĵ���������Ϊ20.10mL��19.90mL��24.00mL����������ȥ������������Һ��ƽ�����Ϊ20.00mL��NaOH��Һ�����Ϊ10.00mL��
HCl��������������NaOH
1 1
0.20molL-1��20.00mL C(NaOH)��10.00mL
��ã�C(NaOH)=0.40mol/L��
��Ʒ��NaOH�����ʵ���Ϊ0.40mol/L��0.5L=0.2mol��
NaOH������Ϊ0.2mol��40g/mol=8.0g����Ʒ����Ϊ8.0g��10.0g��100%=80%��
��4��A����ƿ������ˮϴ����������Һ��ϴ����Ӱ���Һ�������ⶨ�����Ӱ������A������B����ʽ�ζ���δ������ļ���Һ��ϴ��ֱ��ע��������Һ��������Һ��Ũ��ƫС�����V(����)ƫ����c(����)=![]() �������ⶨc(����)ƫ������B������C����ʽ�ζ����ڵζ�ǰ�����ݣ��ζ���������ʧ�����ӵı�Һƫ���������V(����)ƫ����c(����)=
�������ⶨc(����)ƫ������B������C����ʽ�ζ����ڵζ�ǰ�����ݣ��ζ���������ʧ�����ӵı�Һƫ���������V(����)ƫ����c(����)=![]() �������ⶨc(����)ƫ�ͣ���C����D����ȡ�������Һ���ʱ����ʼ���Ӷ������ζ�����ʱ���Ӷ��������V(����)ƫС��c(����)=
�������ⶨc(����)ƫ�ͣ���C����D����ȡ�������Һ���ʱ����ʼ���Ӷ������ζ�����ʱ���Ӷ��������V(����)ƫС��c(����)=![]() �������ⶨc(����)ƫ������D��ȷ���ʴ�ΪD��
�������ⶨc(����)ƫ������D��ȷ���ʴ�ΪD��

 ��У��ʦ������ҵ���Ӻ����Ծ�ϵ�д�
��У��ʦ������ҵ���Ӻ����Ծ�ϵ�д� ȫ�̽��ϵ�д�
ȫ�̽��ϵ�д�����Ŀ��
����ͪ����ɫ��dz��ɫ��Һ�壬��ǿ�ҵĴ̼��Գ�ζ���ܶȣ����ˮ=1����0.95���۵㣺-45�����е㣺155�����ܽ�ȣ�100mL H2O����2.4g��31������
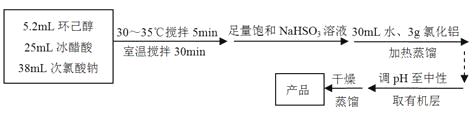
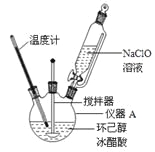
����Ӧ��
�ش��������⣺
��1������A�������� ��
��2���ڷ�Ӧ��ʼ��5min��Ϊ�˽���ϵ�¶�ά����30��35���������ñ�ˮԡ��ȴ�⣬��ȡ�Ĵ�ʩ���� �� ��
��3�����뱥��NaHSO3��Һʱ��������Ҫ��Ӧ�� �������ӷ���ʽ��ʾ����ȷ������ı���NaHSO3��Һ�Ѿ�������ʵ������� ��
��4��Ϊ�����Һ��pH�������ԣ����Լ�����Լ��� ��
A��ϡ���� | B����ˮ̼���� | C��Ũ���� | D���������ƹ��� |
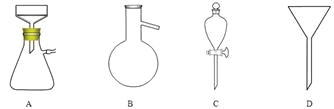
��5������pH������뾫��ʳ�Σ���Ŀ���� ����ȡ�л�����ʱʹ�õ���Ҫ������ ��������ͼ��Ӧ����ĸ����
��6��ʵ���������Ż���ʵ��ɰ�������Ҫ��С���Ա�����б�Ҫ��NaClO��Һ�����Ũ�Ƚ���̽��������������һϵ�в�ͬŨ�ȵ�NaClO��Һ�����õζ����궨�����巽���ǣ�����Һ��ȡ10.00mL NaClO��Һ��500mL����ƿ�ж��ݣ�ȡ25.00mL����ƿ�У���������ϡ�����KI���壨����������0.1000mol/L Na2S2O3����Һ�ζ����յ㣨�õ�����Һ��ָʾ����������ƽ��ʵ��ƽ������Na2S2O3����Һ18.00mL����֪I2+2Na2S2O3==2NaI+Na2S4O6 �����NaClO��Һ��Ũ���� ��