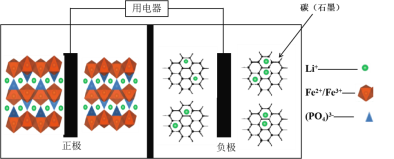
��Ŀ����
����Ŀ��NA�ǰ����ӵ�������ֵ������˵����ȷ���ǣ� ��
A.1 mol Na��O2��ȫ��Ӧ����Na2O��Na2O2�Ļ���ת�Ƶ�����ΪNA������������������Ϊ2NA
B.![]() ��NaHSO3��Һ�У���ˮ�����H+��ĿΪ0.01NA��
��NaHSO3��Һ�У���ˮ�����H+��ĿΪ0.01NA��
C.32.5 gп��һ����Ũ����ǡ����ȫ��Ӧ����������ķ�����Ϊ0.5NA
D.��Ӧ3H2(g)��N2(g)![]() 2NH3(g)����H����92 kJ��mol��1�����ų�����9.2 kJʱת�Ƶ���0.3NA
2NH3(g)����H����92 kJ��mol��1�����ų�����9.2 kJʱת�Ƶ���0.3NA
���𰸡�C
��������
A���ƺ�������Ӧ���Ϊ+1�ۣ���1mol����ȫ��Ӧת��1mol���Ӽ�NA���������ɵIJ��������������ƻ��ǹ������ƣ�����2molNa����1mol�����ӣ���1mol�Ʒ�Ӧ���Ϊ1molNa����������Ӧ�������ӵ����ʵ���Ϊ0.5mol�������������ӹ�1.5NA������A����
B��pH=2��NaHSO3��Һ�У�HSO3���ĵ������ˮ�⣬��Һ�е���������Ҫ������HSO3���ĵ��룬��Һ��ˮ�������������С��0.01NA������B����
C��32.5 gп��һ����Ũ����ǡ����ȫ��Ӧ���������ɶ������������������ɵ������Ϊ0.5mol����������ķ�����Ϊ0.5NA����C��ȷ��
D����Ӧ3H2(g)��N2(g)![]() 2NH3(g)����H����92 kJ��mol��1��ת��6mol���ӣ��ų�����9.2 kJʱ��ת�Ƶ���0.6 NA����D����
2NH3(g)����H����92 kJ��mol��1��ת��6mol���ӣ��ų�����9.2 kJʱ��ת�Ƶ���0.6 NA����D����
��ѡC��

����Ŀ������ʵ���Ҳⶨ����ͭ����ᾧˮ������ʵ�飬��д���пհף�
��1��������������ѡ����������(�ñ����ĸ��д)_____��
A��������ƽ B���в� C��ҩ�� D���ƾ��� E�������� F�������� G��ʯ���� H�������� I������ J�����ż�
�����������⣬����Ҫ��������____��
��2��ijѧ��ʵ���õ��������ݣ�
����ǰ��������g�� | ����ǰ��������g�� | ||
W1�������� | W2������+���壩 | W3������+��ˮ����ͭ�� | |
��һ�� | 15.688 | 17.668 | 16.962 |
��һ�� | 15.688 | 17.744 | 17.002 |
��д���ᾧˮx=___������2λС������ʵ�����=_____%������2λС������
������ѡ����ѡ����ѧ����������ԭ�������(��д��ĸ)____��
A������ǰ����ʱ����δ��ȫ���� B����������Ѿ�����ʧˮ
C�����Ⱥ�����δ�������������ȴ D�����ȹ����о�����������ʧ