��Ŀ����
9��������һ����Ҫ�Ļ���ԭ�ϣ��㷺����ҩ���������� ���Ӻϳɵȹ�ҵ�����ᾧ�����ȵ�100Cʱʧȥ�ᾧˮ����Ϊ��ˮ ���ᣮijѧϰС���ͬѧ���Ը�����Ϊԭ����ˮ��һ����һˮ��ѭ ��������ȡ���ᣮ�ڷ�Ӧ������ˤ1����һ���¶�
���ˢۡ���Ʒ?��
�����������Ϣ�ش��������⣺
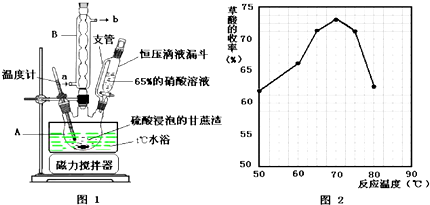
��1��ͼʾ�٢ڵ�����һˮ�����������ͼ1��װ���н��еģ�ָ��װ��B�����������ܣ�Bװ�õ������������������ᣮ
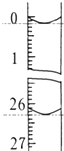
��2��ͼʾ�٢ڵ�����һˮ������У���������������Ӧ��ʱ�����������ͬ������£��� �䷴Ӧ�¶��Կ��췴Ӧ�¶ȶԲ������ʵ�Ӱ�죬�����ͼ2��ʾ����ѡ����ѵķ�Ӧ �¶�Ϊ70�棬Ϊ�˴ﵽͼ2��ʾ���¶ȣ�ѡ��ͼ1��ˮԡ���ȣ����ŵ��DZ��ڿ����¶ȣ�ʹװ�����Ⱦ��ȣ�
��3��Ҫ�ⶨ���ᾧ�壨H2Q04+2H20���Ĵ��ȣ���ȡ7.200g�Ʊ��IJ��ᾧ����������ˮ���250mL��Һ��ȡ25.00mL������Һ����ƿ�У���0.1000mol/L���Ը��������Һ�� ����5H2C2��4+2Mn��4-+6H+=2Mn2++10C02T+8H20����
��ȡ25.00mL������Һ����������ʽ�ζ��ܣ�
���ڲ��ᴿ�Ȳⶨ��ʵ������У����ζ��յ��ȡ�ζ��̶ܿ�ʱ�����ӱ�ҺҺ�棬��ʹʵ����ƫ�ߣ����ƫ�ߡ���ƫ�͡���û��Ӱ�족��
���жϵζ��Ѿ��ﵽ�յ�ķ����ǣ�����ƿ�е������һ�θ�����ر�Һ����ƿ����Һ���dz��ɫ�Ұ���Ӻ�ɫ��
�ܴﵽ�ζ��յ�ʱ�����ĸ��������Һ��20.00mL������ᾧ��Ĵ���Ϊ87.5%��
���� ��1�����������Ľṹ�ص��жϣ���������Ļӷ��Է�����
��2�������¶���������������ж���ѷ�Ӧ�¶ȣ����ݼ��ȵ�Ҫ��ѡ����ȵķ�����
��3���ٸ��ݲ�����Һ������Լ���ȡ��Һ�ľ�ȷ��ѡ����Ӧ��������
�����Ӷ����������ƫ��
���������Ը��������Һ���Ϻ�ɫ�����Եζ��յ���ж�����������ƿ�е������һ�θ�����ر�Һ����ƿ����Һ���dz��ɫ�Ұ���Ӻ�ɫ��
�ܷ�Ӧ�����ĸ�����ص����ʵ�����0.02L��0.1000mol/L=0.002000mol�����Ը��ݷ���ʽ5H2C2O4+2MnO4-+6H+=2Mn2++10CO2��+8H2O��֪�����IJ�������ʵ�����0.005000mol��
��� �⣺��1������װ�õĽṹ�ص��֪��BӦ���������ܣ�����������лӷ��ԣ������ӷ�������������������������������ã�
�ʴ�Ϊ�������ܣ������������
��2������ͼ3��֪�����¶�Ϊ70��ʱ�������������ߣ�����ѡ�����ѷ�Ӧ�¶���70�森�����¶Ȳ�����100�棬���Բ���ˮԡ���ȣ���������ˮԡ����ʱ���ڿ����¶ȣ�����ʹװ�����Ⱦ��ȣ�
�ʴ�Ϊ��70�棻���ڿ����¶ȣ�ʹװ�����Ⱦ��ȣ�
��3���ٲ�����Һ�����ԣ�������ȡ25.00mL������Һ����������ʽ�ζ��ܣ�
�ʴ�Ϊ����ʽ�ζ��ܣ�
�ڵζ��յ��ȡ�ζ��̶ܿ�ʱ�����ӱ�ҺҺ�棬���ڶ������϶������������Զ���ƫ�������ĵĸ�����ص����ƫ����������ʵ���ƫ��ʹʵ����ƫ�ߣ���D��ȷ��
�ʴ�Ϊ��ƫ�ߣ�
���������Ը��������Һ���Ϻ�ɫ�����Եζ��յ���ж�����������ƿ�е������һ�θ�����ر�Һ����ƿ����Һ���dz��ɫ�Ұ���Ӻ�ɫ��
�ʴ�Ϊ������ƿ�е������һ�θ�����ر�Һ����ƿ����Һ���dz��ɫ�Ұ���Ӻ�ɫ��
�ܷ�Ӧ�����ĸ�����ص����ʵ�����0.02L��0.1000mol/L=0.002000mol�����Ը��ݷ���ʽ5H2C2O4+2MnO4-+6H+=2Mn2++10CO2��+8H2O��֪�����IJ�������ʵ�����0.005000mol�����ԭ��Ʒ�в�������ʵ�����0.005000mol��$\frac{250mL}{25mL}$=0.05000mol/L��������=0.05000mol/L��126g/mol=6.3g��
���Բ���Ĵ����� $\frac{6.3g}{7.2g}$��100%=87.5%��
�ʴ�Ϊ��87.5%��
���� ���⿼����������ɡ�������ʵ��̽��������ʵ������жϣ��ζ�ʵ��ע������ͼ���Ӧ�ã����ջ����ǽ���ؼ�����Ŀ�Ѷ��еȣ������ڿ���ѧ���ķ������⡢������������Լ����ݴ���������

 �¿α�ͬ��ѵ��ϵ�д�
�¿α�ͬ��ѵ��ϵ�д� һ����ʦ����Ӧ����������һ��ȫϵ�д�
һ����ʦ����Ӧ����������һ��ȫϵ�д�| A�� | �������ơ��������ơ������İ���ͭ[Cu��NH3��4]SO4���⻯�� | |
| B�� | ����李��⻯李���������������[Ag��NH3��2]OH������� | |
| C�� | ���ᡢ�������ơ������ơ��������� | |
| D�� | �Ȼ�李��������ơ�˫��ˮ���������� |
 ijѧ��������֪���ʵ���Ũ�ȵ��������ⶨδ֪���ʵ���Ũ�ȵ�NaOH��Һʱ��ѡ���̪��ָʾ��������д���пհף�
ijѧ��������֪���ʵ���Ũ�ȵ��������ⶨδ֪���ʵ���Ũ�ȵ�NaOH��Һʱ��ѡ���̪��ָʾ��������д���пհף���1���жϵζ��յ��������Һǡ����dz��ɫ�����ɫ���Ұ�����ڲ��ָ���
��2�����в����п���ʹ����NaOH��Һ��Ũ����ֵƫ�͵���D
A����ʽ�ζ���δ�ñ�������ϴ��ֱ��ע�������
B���ζ�ǰʢ��NaOH��Һ����ƿ������ˮϴ����û�и���
C����ʽ�ζ����ڵζ�ǰ�����ݣ��ζ���������ʧ
D����ȡ�������ʱ����ʼ���Ӷ������ζ�����ʱ���Ӷ���
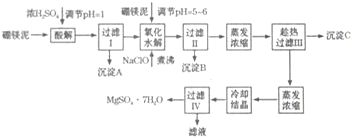
��3�����ζ���ʼ�ͽ���ʱ����ʽ�ζ����е�Һ����ͼ��ʾ��������������Һ�����Ϊ26.10ml��
��4��ijѧ������3��ʵ��ֱ��¼�й����������
| �ζ� ���� | ����NaOH��Һ�����/mL | 0.1000mol/L��������/mL | |
| �ζ�ǰ�̶� | �ζ���̶� | ||
| ��һ�� | 25.00 | 0.00 | 26.15 |
| �ڶ��� | 25.00 | 0.56 | 30.30 |
| ������ | 25.00 | 0.20 | 26.35 |
�������ϱ����ݼ����NaOH��Һ�����ʵ���Ũ��0.1046mol/l ��������ȡ4λ��Ч������
��1��������������������������ʽ��ȫ����ʱ��pH
| ���� | Al��OH��3 | Fe��OH��3 | Fe��OH��2 | Mg��OH��2 |
| pH | 5.2 | 3.2 | 9.7 | 11.2 |
| �¶�/�� | 10 | 30 | 40 | 50 | 60 |
| CaSO4 | 0.19 | 0.21 | 0.21 | 0.20 | 0.19 |
| MgSO4•7H2O | 30.9 | 35.5 | 40.8 | 45.6 | -- |
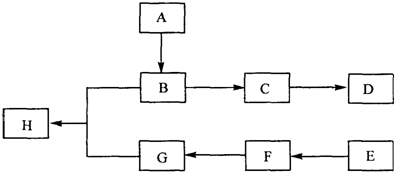
������������ͼ���ο�����pH���ܽ�����ݣ��Իش��������⣺
��1����þ�����Ũ����ʱ��FeO�����ı仯��bc��ѡ����ţ���
a�����ۻ� b�������� c�����ܽ�
��2������1����Һ�м�����þ�࣬������Һ��pH=5-6���ټ���NaCLO��Һ������У���Ŀ���У��ٽ���Һ�е�Mn2+������MnO2���÷�Ӧ�����ӷ���ʽΪMn2++ClO-+H2O=MnO2��+2H++Cl-���ڽ�Fe2+����ΪFe3+���÷�Ӧ�����ӷ���ʽΪ6Fe2++3ClO-+3H2O=2Fe��OH��3��+4Fe3++3Cl-��2Fe2++ClO-+2H+=2Fe3++Cl-+H2O��
��3������B�г�MnO2��SiO2�����Fe��OH��3��Al��OH��3�����ʣ��ѧʽ��
��4������C�Ļ�ѧʽ��CaSO4•2H2O��CaSO4�����ˢ�����ȹ��˵������Ƿ�ֹMgSO4���¶Ƚ���ʱ�ᾧ������
| A�� | ԭ�Ӱ뾶��A��B��D��C | B�� | ԭ��������d��c��b��a | ||
| C�� | ���Ӱ뾶��C��D��B��A | D�� | ���ʵĻ�ԭ�ԡ�A��B��D��C |
 ��
��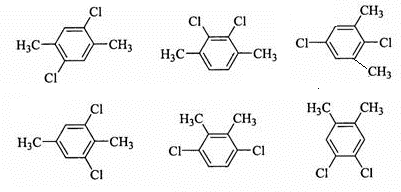 ���������֣����������֣�
���������֣����������֣�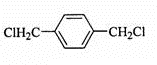
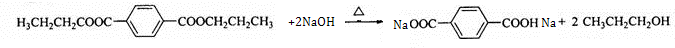 ���÷�Ӧ�ķ�Ӧ������ȡ����Ӧ��
���÷�Ӧ�ķ�Ӧ������ȡ����Ӧ��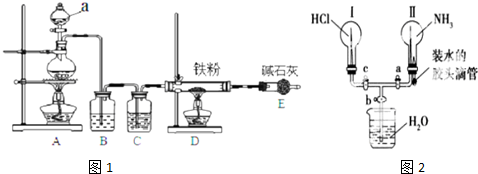
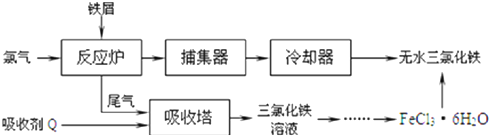
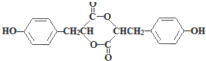 ����·������ͼ��ʾ��
����·������ͼ��ʾ��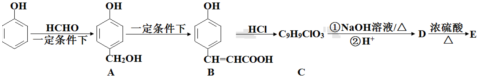
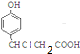 +3NaOH$\stackrel{��}{��}$$\stackrel{��}{��}$$\stackrel{��}{��}$
+3NaOH$\stackrel{��}{��}$$\stackrel{��}{��}$$\stackrel{��}{��}$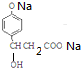 +NaCl+2H2O��
+NaCl+2H2O�� 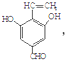 ��д�������ͬ���칹��Ľṹ��ʽ
��д�������ͬ���칹��Ľṹ��ʽ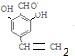 ��
��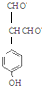 ��
��