��Ŀ����
7��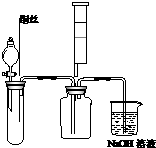 ��1��ijͬѧΪ��֤�����۲쵽ͭ��ϡ���ᷴӦ�IJ���NO��ʵ��ʱ��������ͼ�Թ��м���һ�����Ŀ�״���壬�ٵμӹ�����ϡ���ᣬ����Ӧ��������������Թܺ��ٽ�ͭ˿����ϡ������Һ�У�����Ϊ�Թ��еĿ�״������̼��ƣ��������Ǹ����Թ��еĿ�����ʵ�����ʱ����ע�������Թ����������������������Ŀ���Ǽ���NO���۲쵽����ɫ���壮
��1��ijͬѧΪ��֤�����۲쵽ͭ��ϡ���ᷴӦ�IJ���NO��ʵ��ʱ��������ͼ�Թ��м���һ�����Ŀ�״���壬�ٵμӹ�����ϡ���ᣬ����Ӧ��������������Թܺ��ٽ�ͭ˿����ϡ������Һ�У�����Ϊ�Թ��еĿ�״������̼��ƣ��������Ǹ����Թ��еĿ�����ʵ�����ʱ����ע�������Թ����������������������Ŀ���Ǽ���NO���۲쵽����ɫ���壮��2��ͭƬ��Ũ���ᷴӦ��������Һ����ɫ��Cu2+��NO2��������ɫ����ϡ��Ľ��������ΪҪ֤���������ۣ��ɲ�ȡ�����ʵ�鷽��Ӧ�Ǽ��ȣ��۲쵽���������к���ɫ�����������Һ����ɫ��
���� ��1��֤�����۲쵽ͭ��ϡ���ᷴӦ�IJ���NO����ͼ��֪���Թ���Ϊ̼��ƣ����Һ©���е����ᷴӦ���ɶ�����̼����װ���ڿ����ų�����ֹCu�����ᷴӦ���ɵ�NO��������ʵ�����ʱ����ע�������Թ����������������������NO���������ɶ���������
��2��NO2Ϊ����ɫ���壬���ȿ��ݳ���
��� �⣺��1����֤NO�����ɣ�Ӧ��ֹ�䱻���������Թ��еĿ�״������̼��ƣ�������Ϊ�����Թ��еĿ�����ʵ�����ʱ����ע�������Թ����������������������Ŀ���Ǽ���NO���۲쵽����ɫ���壬
�ʴ�Ϊ��̼��ƣ������Թ��еĿ���������NO���۲쵽����ɫ���壻
��2��������Һ����ɫ��Cu2+��NO2��������ɫ����ϡ��Ľ����Ҫ֤���������ۣ��ɲ�ȡ�����ʵ�鷽��Ӧ�Ǽ��ȣ��۲쵽�к���ɫ�����������Һ����ɫ��
�ʴ�Ϊ�����ȣ��к���ɫ�����������Һ����ɫ��
���� ���⿼�����ʵ��Ʊ�ʵ�飬Ϊ��Ƶ���㣬����NO���Ʊ�ԭ����NO�����ʡ�ʵ��װ�õ�����Ϊ���Ĺؼ������ط�����ʵ�������Ŀ��飬��Ŀ�ѶȲ���

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ
17��NA��ʾ�����ӵ�������ֵ������˵����ȷ���ǣ�������
| A�� | һ�������£�1 mol N2��3 mol H2��ַ�Ӧ���������е�N-H����ĿΪ6NA | |
| B�� | ��ȫȼ��1.5molCH3CH2OH��C2H4�Ļ���ת�Ƶ�����Ϊ18NA | |
| C�� | 100 g 98%��Ũ�����к���ԭ�Ӹ���Ϊ4NA | |
| D�� | 1 L 0.1 mol/L��Na2S��Һ��S2-��HS-������Ϊ0.1NA |
15�����з�Ӧ������CO2���������ǣ�������
| A�� | O2+C$\frac{\underline{\;����\;}}{\;}$CO2 | B�� | 2NaOH+CO2�TNa2CO3+H2O | ||
| C�� | 2Na2O2+CO2�T2Na2CO3+O2 | D�� | 2Mg+CO2$\frac{\underline{\;��ȼ\;}}{\;}$2MgO+C |
2��0.1molij��������������Ӧʱ���������������ԭ��������������1.6g����˽��������ǣ�������
| A�� | Na | B�� | Cu | C�� | Fe | D�� | Al |
19��ԭ����������11-35֮�����������Ԫ��A��B�����ӷֱ�ΪAm+��Bn-����д�±�
| λ�� | A��B��������� | ���Ӱ뾶�Ƚ� | ||||
| ���� | ���� | ���� | ||||
| Am+��Bn-����������Ӳ� | A | |||||
| B | ||||||
| Am+��Bn-���һ�����Ӳ� | A | |||||
| B | ||||||
| Am+��Bn-���Ӳ�ṹ��ͬ | A | |||||
| B | ||||||
16����9.0gͭ�����Ļ����Ͷ��100mlϡ�����г�ַ�Ӧ�õ������1.12LNO��ʣ��4.8g��������������100ml��Ũ�ȵ�ϡ���������ȫ�ܽ⣬�ֵõ������1.12LNO������Ӧ�����Һ�е���KSCN��Һ����죬������˵����ȷ���ǣ�������
| A�� | ��һ����100mlϡ���ᷴӦʣ4.8g����ΪCu��Fe | |
| B�� | ��Ӧǰϡ��������ʵ�����Ũ��Ϊ1.5 mol•L-1 | |
| C�� | �����������������Һ���ټ���������ϡ���ỹ�ɵõ������0.56LNO | |
| D�� | ԭ�������Cu��Fe�����ʵ����ֱ�Ϊ0.06mol��0.09mol |
9���������ǿ�Ⱥܸߣ���������ѹ�����裬�����������Ӳ������֮һ���������մɿ���ȼ���ֻ���ȼ���ҡ���е�ܷ��������Һ�ĵ�űõĹܵ������š�������ģ�ߡ���ˮ���뻷�ȣ��Ʊ�������ɽ���۷��ڵ����м�����1000������ֱ�ӵ����Ƶã�ʵ�����Ʊ�������װ��ʾ��ͼ���£�

�ش��������⣺
��1��a�����������Ƿ�Һ©��
��2��д��NaNO2�ͣ�NH4��2SO4��Ӧ�Ʊ������Ļ�ѧ����ʽ2NaNO2+��NH4��2SO4$\frac{\underline{\;\;��\;\;}}{\;}$2N2��+Na2SO4+4H2O
��3��װ��B�������dz�ȥ�����������������װ��C�������dz�ȥˮ������
��4����֪������Ļ�ѧ�����ȶ������������������������Һ����һ�ְ�ɫ�Ľ�״������һ���Σ���д���˻�ѧ��Ӧ����ʽ��Si3N4+4HF+9H2O=3H2SiO3��+4NH4F��
��5��ʵ������ijЩ�������ȡ���ռ���β������װ������ͼ��ʾ��ʡ�Լгֺ;���װ�ã������ô�װ�úͱ����ṩ������������ʵ�飬�������ѡ����D
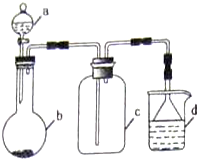

�ش��������⣺
��1��a�����������Ƿ�Һ©��
��2��д��NaNO2�ͣ�NH4��2SO4��Ӧ�Ʊ������Ļ�ѧ����ʽ2NaNO2+��NH4��2SO4$\frac{\underline{\;\;��\;\;}}{\;}$2N2��+Na2SO4+4H2O
��3��װ��B�������dz�ȥ�����������������װ��C�������dz�ȥˮ������
��4����֪������Ļ�ѧ�����ȶ������������������������Һ����һ�ְ�ɫ�Ľ�״������һ���Σ���д���˻�ѧ��Ӧ����ʽ��Si3N4+4HF+9H2O=3H2SiO3��+4NH4F��
��5��ʵ������ijЩ�������ȡ���ռ���β������װ������ͼ��ʾ��ʡ�Լгֺ;���װ�ã������ô�װ�úͱ����ṩ������������ʵ�飬�������ѡ����D

| ѡ�� | a���� | b���� | c�ռ������� | d�е����� |
| A | Ũ��ˮ | NaOH | NH3 | H2O |
| B | Ũ���� | Na2SO3 | SO2 | ����ʯ��ˮ |
| C | ϡ���� | Fe | NO2 | H2O |
| D | Ũ���� | KMnO4 | Cl2 | NaOH��Һ |
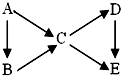 AΪ���ʣ�B��C��D��EΪ��A������ͬԪ�صĻ��������֮��������ת����ϵ��
AΪ���ʣ�B��C��D��EΪ��A������ͬԪ�صĻ��������֮��������ת����ϵ��