��Ŀ����
��ѧ����������������Ϊ�Ҵ���ˮ����ϩ�Ĵ������������ʵ�顣���ұ���ʾ�����ͷ�Ӧ����������ƿ�м���һ����P2O5����ע��95%���Ҵ��������ȣ��۲�����
| ʵ�� | P2O5 /g | 95%�Ҵ���/ mL | ���ȷ�ʽ |
| ʵ��1 | 2 | 4 | �ƾ��� |
| ʵ��2 | 2 | 4 | ˮԡ70�� |

ʵ�������£�
| ʵ�� | ʵ������ | ||
| ����ƿ | �ռ�ƿ | �Թ� | |
| ʵ��1 | �ƾ�����ʱ�����̷ų������İ�������ʼ�����ݲ��������þƾ��Ƽ���ʱ�����ݼӿ����ɲ����ڣ�����ճ��״Һ�塣 | ����ɫҺ�� | ��Һ��ɫ |
| ʵ��2 | �ƾ�����ʱ���������������ɣ�����ˮԡ����ʱ�����������ݣ���Ӧһ��Сʱ����Ӧƿ������ճ��״Һ�� | ����ɫҺ�� | ��Һ����ɫ |
�����������ϣ����������ա�
��1��д���Ҵ�����ϩ�Ļ�ѧ����ʽ ��
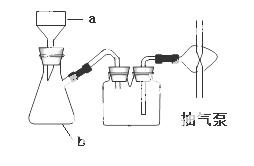
��2����ͼװ���������ܵ�������___________����ˮ��Ϊ���a����b����______��Ũ�������
���� ��
��3��ʵ��1ʹ������Ȼ�̼��Һ��ɫ��������___________��
��4��ʵ��2�У�ˮԡ�������������� �� �����ȡ��г�������ʯ�������⣩��
��5������ƿ���ռ�ƿ�е�Һ�徭����Ϊ������������д������ƿ����������Ļ�ѧ����ʽ
_____________________��P2O5��ʵ��1�е�������______________________ ��
��6������ʵ��1��2�����ƶϣ�
����P2O5��Ϊ���������ϩ�ķ�Ӧ������____________
��P2O5��95%�Ҵ��ڼ��������¿��Է������л���Ӧ��������________��Ӧ��
�����12�֣�ÿ��1��,38��2�֣�39�ڶ���2�֣�
��1��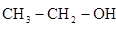
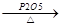
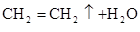
��2������������ b����ˮ
��3����ϩ
��4�����ձ����¶ȼ�
��5�� ����Ӧ��ʹ���
����Ӧ��ʹ���
��6���ٽϸ��¶��»�ֱ�Ӽ��ȡ���������Ӧ��
���������������1���Ҵ���P2O5�����������·�����ȥ��Ӧ������ϩ��ˮ��
��2���������������͵��������ã�ˮ�������������෴������Ч���ã�����Y���·���bΪ��ˮ�ڣ����ɵ���ϩ�к���ˮ�֣�Ũ���������ˮ�����á�
��3����ϩ��Br2�����ӳɷ�Ӧ���Ӷ�ʹ������Ȼ�̼��Һ��ɫ��
��4��ˮ��Ҫ���ձ�ʢ�ţ�Ϊ�˲���ˮԡ���¶���Ҫ�¶ȼơ�
��5��P2O5��ˮ��Ӧ��������Ҵ�������ȥ��Ӧʱ��P2O5�������ã�P2O5�������ɵ�H2O��Ӧ����Ӧ������á�
��6���ٵ��þƾ��Ƽ���ʱ�����ݼӿ����ɲ����ڣ�����ճ��״Һ�壬˵�������ϩ�ķ�Ӧ�����ǽϸ��¶��»�ֱ�Ӽ��ȡ�
��P2O5��ˮ��Ӧ����H3PO4��H3PO4���Ҵ��ɷ���������Ӧ��
���㣺���⿼���л���ѧ����ʽ����д������������ʹ�ü����á��Լ������úͷ�Ӧ������ѡ��

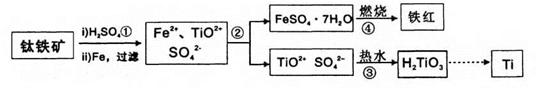
����ֲ���纣���������к��зḻ�ĵ�Ԫ�أ���Ԫ���Ե����ӵ���ʽ���ڡ�ʵ������Ӻ�������ȡ����������£�
��1��ָ�������۵����ƣ� ���������г�������Cl2��Ŀ���� ��
��2����ȡ��Ĺ����У��ɹ�ѡ����Լ��� ( )
| A���ƾ� | B�����Ȼ�̼ | C������ | D������ |

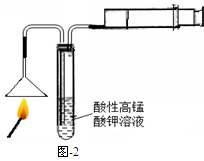
��3���Ӻ�����л���Һ����ȡ��ͻ����л��ܼ����辭������ͼI��ijͬѧ��Ƶ�����װ�ã�ͼ�����ԵĴ����� ��
��4����ͬѧ��Ϊ����ʱ���ʹ��ˮԡ���ȣ�ʹ��ˮԡ���ŵ��ǣ� ����������������ۼ��� �����������ƣ��
��5�� ʵ��۷�������ķ�Һ�к���Cl�C��SO42�C����ֻȡһ����Һ����μ����Cl�C��SO42�C�����μ����Լ���Ϊ�� �� ��
��6��ijС��ͬѧʵ��ʱ���õ�һ�����ʵ���Ũ�ȵĵ�ˮ��Һ225mL������ʱ��Ҫ����������ƽ�����������ձ��⣬���� �� ����ҡ��ʱ������Һ����ڿ̶��ߣ��������ҺŨ������ƫ��ƫС����Ӱ�죩��
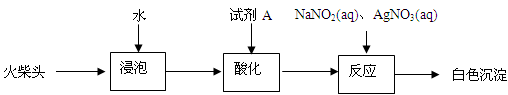
ijС�������ͼװ�ã����ּг�װ������ȥ������̽����ʪ��Cl2��Na2CO3��Ӧ�õ��������ʵijɷ֡�
��1���Լ�X�Ļ�ѧʽ ��A����ȡCl2�����ӷ���ʽ ��
��2���Լ�Y������Ϊ ��
��3��ͨ��һ������ʪ��Cl2��Ӧ����⣬D��ֻ��Cl2Oһ�����壬C��ֻ��һ�������⣬ͬʱ����NaHCO3�ȣ�ijͬѧ��C�����ù�������ijɷֽ���̽����
������������衣
����1���������ֳɷ֣�NaHCO3�� ��
����2���������ֳɷ֣�NaHCO3�� ��
����Ʒ���������ʵ�顣д��ʵ�鲽���Լ�Ԥ������ͽ��ۡ�
��ѡʵ���Լ�������������ˮ��ϡHNO3��BaCl2��Һ������ʯ��ˮ��AgNO3��Һ���Թܡ�С�ձ���
| ʵ�鲽�� | Ԥ������ͽ��� |
 ����1��ȡC�е�����������Ʒ���Թ��У��μ���������ˮ�������ܽ⣬Ȼ��������Һ�ֱ�����A��B�Թ��С� ����1��ȡC�е�����������Ʒ���Թ��У��μ���������ˮ�������ܽ⣬Ȼ��������Һ�ֱ�����A��B�Թ��С� | |
| ����2����A�Թ��еμ�BaCl2��Һ | |
| ����3�� | |
��4����֪��Cl2O��ˮ��Ӧ���ɴ����ᣬ��D��Cl2O����E��Ӧ�Ļ�ѧ����ʽΪ��
��
��(Sr)Ϊ�������ڢ�A��Ԫ�ء��ߴ���ˮ�Ȼ��Ⱦ���(SrCl2?6H2O)���кܸߵľ��ü�ֵ��61��ʱ���忪ʼʧȥ�ᾧˮ��100��ʱʧȥȫ���ᾧˮ���ù�ҵ̼���ȷ�ĩ(������Ba��Fe�Ļ�����)�Ʊ��ߴ���ˮ�Ȼ��ȵĹ�������ͼ��
��ش�
��1����������30%H2O2������ �������ӷ���ʽ��ʾ����
��2��������������ȷ�ĩ�������� ����ҵ����50��60���ȷ紵����ˮ�Ȼ��ȣ�ѡ����¶ȵ�ԭ���� ��
��3������ܽ��е�ʵ�����Ϊ �� ��������У�ϴ���Ȼ��Ⱦ������ѡ�� ��
| A��ˮ | B������ | C������������Һ | D���Ȼ��ȱ�����Һ |
��5������ԭ��ҵ̼���ȷ�ĩ��̼���ȵ��������� ���м���ʽ���ɣ���
�����������ų������ı���[��Ҫ��BaCO3��BaSO3��Ba(FeO2)2��]��ij��Ҫ����BaCO3�Ļ��������ñ�����ȡBa(NO3)2���弰����������䲿�ֹ����������£�
��֪���� Fe(OH)3��Fe(OH)2��ȫ����ʱ����Һ��pH�ֱ�Ϊ3.2��9.7
�� Ba(NO3)2����ˮ���ܽ�Ƚϴ�����ˮ���ܽ�Ƚ�С
�� KSP(BaSO4)=1.1��10��10��KSP (BaCO3)=5.1��10��9
��1���ó�������BaCO3��������BaSO4���������ᴿ�ķ����ǣ�����Ʒ���������ı���Na2CO3��Һ�У���ֽ��裬���ˣ�ϴ�ӡ������ӷ���ʽ˵���ᴿԭ���� ��
��2��������������ʱ��Ba(FeO2)2��HNO3��Ӧ�������������Σ���ѧ����ʽΪ��
��
��3���ó���ϱ���ʵ�ʣ�ѡ�õ�XΪ ��
| A��BaCl2 | B��BaCO3 | C��Ba(NO3)2 | D��Ba(OH)2 |
��5������IIIΪ ��
��6������3���ĸҺӦѭ�������� �С����a������b����c����
��7����ȡw�˾�����������ˮ���������������ᣬ��ַ�Ӧ���ˡ�ϴ�ӡ����������������Ϊm�ˣ����Ba(NO3)2�Ĵ���Ϊ ����Է���������Ba(NO3)2Ϊ261��BaSO4Ϊ233����
������ˮԡ���ȵ�ʵ���ǣ� ��
| A������������Ӧ | B��������Ӧ | C���Ʒ�ȩ��֬ | D�����Ҵ�����ϩ |