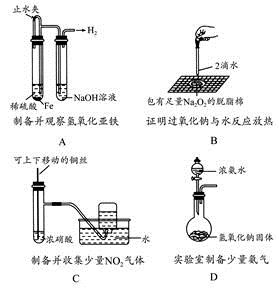
��Ŀ����
ij��ѧС��ͬѧΪ����֤�ճ��������û��ͷ�еĻ�ѧ�ɷ֣���KClO3��MnO2��S�ȣ������������ʵ�����̣���ͼ-1���� 
�Իش��������⣺
��1��ȼ�ŵĻ��ͼ-2����ʵ�飬���Թ����ܹ۲쵽 ���������֤�����ͷ�к�����Ԫ�ء�ͼ����Ͳ�������� ��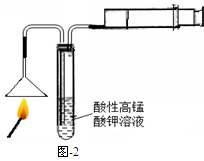
��2��Ϊ��֤�����ͷ�к�����Ԫ�أ�������ʵ�鲽���� ��
��3����ͬѧ���������ͷ��KClO3����һʵ�鷽����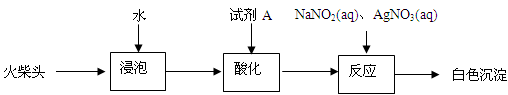
�Լ�AΪ ������NaNO2��Ŀ���� ��
��4�����ʵ�飺������֤����D�к���MnO2��һ��ʵ�鷽������д���йط�Ӧ�Ļ�ѧ����ʽ�� ��
��1��KMnO4��Һ��ɫ����ɫ��dz��������ȷ������2�֣���
����˼��ȷ���ɣ���1�֣�
��2��ȡ������ҺC�����Թ��У��Ⱥ�����HNO3��AgNO3��Һ�����۲쵽��ɫ����������������֤�����ͷ�к�����Ԫ�ء���1�֡�3��
��3�������HNO3��1�֣�����ԭKClO3��1�֣�
��4��ȡ��������D����װ������H2O2���Թ��У��д������ݷų�˵������MnO2��2�֣���
2H2O2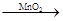 2H2O+O2����2�֣���������ȷ�𰸡�
2H2O+O2����2�֣���������ȷ�𰸡�
���������������1��������Ԫ��ȼ���������˶������������ʹKMnO4��Һ��ɫ����ɫ��dz������Ͳ����ʹȼ�ղ���������˳�������Թܡ�
��2����Ԫ��ȼ������Ҫ�������Ȼ������壬ֻҪȡ������ҺC�����Թ��У��Ⱥ�����HNO3��AgNO3��Һ�����۲쵽��ɫ����������������֤�����ͷ�к�����Ԫ�ء��൱���������ӵļ��顣
��3��KClO3��ǿ�������ԣ������Ի����£���������HNO3��Ҫ���ữ�������������н�ǿ�Ļ�ԭ�ԣ����Ի�ԭKClO3Ϊ�����ӡ�
��4����֤�Ƿ���MnO2��������ʵ������ȡ�����ķ�����Ҳ������Ϊ����ȡ��������D����װ������H2O2���Թ��У��д������ݷų�˵������MnO2����ط�Ӧ 2H2O2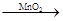 2H2O+O2��
2H2O+O2��
���㣺������ʵ��̽����˼·��Ԫ�ػ������еĻ���֪ʶ��

 �п�������㾫��ϵ�д�
�п�������㾫��ϵ�д� ������ĩ��ϰ��ѵ��ϵ�д�
������ĩ��ϰ��ѵ��ϵ�д� С��ʿ��ĩ����100��ϵ�д�
С��ʿ��ĩ����100��ϵ�д���Ƴ������������ƹ��գ������ŷŵķ�ˮ�к��еľ綾CN-���ӣ����������ƹ�����������������Ƶķ�ˮʱ�����ڴ���TiO2�����£�����NaClO��CN-����������OCN-���������������¼�����NaClO������N2��CO2������������Ա���ܱ�ϵͳ������ͼװ�ý���ʵ�飬��֤��������������Ч�ԣ����ⶨCN-�������İٷ��ʡ���Ũ����CN-���ӵ���ˮ�����NaClO��Һ�Ļ��Һ��200mL������CN-��Ũ��Ϊ0.05mol��L-1��������У�������Ƥ����һ��ʱ�����Ƥ���ͻ�����ʹ��Һȫ���������У��رջ������ش��������⣺
��1�����з�Ӧ�����ӷ���ʽΪ ��
��2���������ɵ������N2��CO2�⣬���и�����HCl��Cl2�ȣ�����ʵ����ͨ���ⶨ������̼������ȷ����CN-�Ĵ���Ч��������м���ij����Լ���___________������ĸ��
| A������ʳ��ˮ | B������NaHCO3��Һ | C��ŨNaOH��Һ | D��Ũ���� |
װ�м�ʯ�ҵĸ���ܵ������� ��
��4������ʢ�к�Ca(OH)2 0.02mol��ʯ��ˮ����ʵ�������й�����0.82 g���������ʵ���в��CN-�������İٷ��ʵ��� �����ò��ֵ��ʵ�ʴ����İٷ������ƫ�ͣ����Ҫ˵�����ܵ�����һ��ԭ�� ��
��5�������һ�������ȷ�ȵĽ��飨Ҫ�пɲ����ԣ�����ʹ������ù��ڸ��ӣ� ��
��ѧ����������������Ϊ�Ҵ���ˮ����ϩ�Ĵ������������ʵ�顣���ұ���ʾ�����ͷ�Ӧ����������ƿ�м���һ����P2O5����ע��95%���Ҵ��������ȣ��۲�����
| ʵ�� | P2O5 /g | 95%�Ҵ���/ mL | ���ȷ�ʽ |
| ʵ��1 | 2 | 4 | �ƾ��� |
| ʵ��2 | 2 | 4 | ˮԡ70�� |

ʵ�������£�
| ʵ�� | ʵ������ | ||
| ����ƿ | �ռ�ƿ | �Թ� | |
| ʵ��1 | �ƾ�����ʱ�����̷ų������İ�������ʼ�����ݲ��������þƾ��Ƽ���ʱ�����ݼӿ����ɲ����ڣ�����ճ��״Һ�塣 | ����ɫҺ�� | ��Һ��ɫ |
| ʵ��2 | �ƾ�����ʱ���������������ɣ�����ˮԡ����ʱ�����������ݣ���Ӧһ��Сʱ����Ӧƿ������ճ��״Һ�� | ����ɫҺ�� | ��Һ����ɫ |
�����������ϣ����������ա�
��1��д���Ҵ�����ϩ�Ļ�ѧ����ʽ ��
��2����ͼװ���������ܵ�������___________����ˮ��Ϊ���a����b����______��Ũ�������
���� ��
��3��ʵ��1ʹ������Ȼ�̼��Һ��ɫ��������___________��
��4��ʵ��2�У�ˮԡ�������������� �� �����ȡ��г�������ʯ�������⣩��
��5������ƿ���ռ�ƿ�е�Һ�徭����Ϊ������������д������ƿ����������Ļ�ѧ����ʽ
_____________________��P2O5��ʵ��1�е�������______________________ ��
��6������ʵ��1��2�����ƶϣ�
����P2O5��Ϊ���������ϩ�ķ�Ӧ������____________
��P2O5��95%�Ҵ��ڼ��������¿��Է������л���Ӧ��������________��Ӧ��
ʵ�����Թ�ҵ̼��ƣ�������Na+��Al3+��Fe3+�����ʣ�Ϊԭ����ȡCaCl2��H2O��CaO2����Ҫ�������£�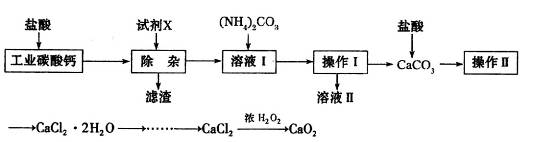
��1�������Լ�X��������ҺpHΪ���Ի������Գ�ȥ��Һ��Al3+��Fe3+����������Ҫ�ɷ���___________���Լ�X����ѡ�����е�________�����ţ���
| A��CaO | B��CaCO3 | C��NH3��H2O | D��Ba(OH)2 |
��3����CaCl2��ȡCaO2�ķ�Ӧ�У��¶Ȳ���̫�ߵ�ԭ����_______________��
��4��������װ�òⶨ��ҵ̼��Ƶ���������
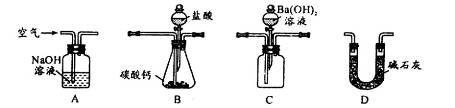
�ټ���װ��B���������õ�ʵ�������__________________________��
�ڰ�A��B��C��D˳�����ӣ�Ȼ���Aװ��ͨ�������Ŀ����_______________��
��װ��D������Ϊ______________________��
��ʵ��ʱ��ȷ��ȡ10.00g��ҵ̼���3�ݣ�����3�βⶨ�����BaCO3������ƽ������Ϊ17.73g������Ʒ��CaCO3����������Ϊ__________________��
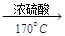 CH2=CH2����H2O������Ũ�������ǿ�����ԣ��丱�����ж�������Ͷ�����̼�ȡ�ijͬѧ����������Ϣ��ʵ��Ŀ��ѡ������ʵ��װ�����ʵ��̽��(ÿ��װ�ö������ɸ�)��
CH2=CH2����H2O������Ũ�������ǿ�����ԣ��丱�����ж�������Ͷ�����̼�ȡ�ijͬѧ����������Ϣ��ʵ��Ŀ��ѡ������ʵ��װ�����ʵ��̽��(ÿ��װ�ö������ɸ�)��