��Ŀ����
���������(NH2COONH4)��һ�ְ�ɫ���壬�ֽ⡢��ˮ�⣬���������ϡ�������ϴ�Ӽ��ȡ�ij��ѧ��ȤС��ģ�ҵԭ���Ʊ���������泥���Ӧ�Ļ�ѧ����ʽ���£�2 NH3(g)+CO2(g)  NH2COONH4(s) ��H��0
NH2COONH4(s) ��H��0
��1��ʵ�����Ʊ�NH3�Ļ�ѧ����ʽ�ǣ� ��
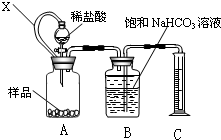
��2���Ʊ���������淋�װ������ͼ��ʾ���Ѱ����Ͷ�����̼ͨ�����Ȼ�̼�У����Ͻ����ϣ����ɵİ��������С�������������Ȼ�̼�С���������϶�ʱ��ֹͣ�Ʊ���

ע�����Ȼ�̼��Һ��ʯ����Ϊ���Խ��ʡ�
�ٷ������ñ�ˮ��ȴ��ԭ���� ��Һ��ʯ������ƿ�������� ��
�ڴӷ�Ӧ��Ļ�����з������Ʒ��ʵ�鷽���� (��д��������)��Ϊ�˵õ������Ʒ��Ӧ��ȡ�ķ����� (��дѡ�����)��
a. ��ѹ���Ⱥ�� b. ��ѹ���Ⱥ�� c. ���40 �����º��
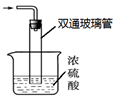
��β������װ������ͼ��ʾ��˫ͨ�����ܵ����ã� ��
Ũ��������ã� �� �� ����������������
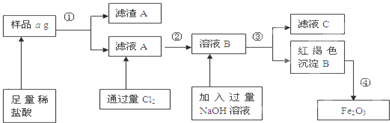
��3��ȡ�ֱ��ʶ�����̼����淋İ����������Ʒ0.7825 g��������ʯ��ˮ��ִ�����ʹ̼Ԫ����ȫת��Ϊ̼��ƣ����ˡ�ϴ�ӡ�����������Ϊ1.000 g������Ʒ�а�������淋����ʵ�������Ϊ ��
 NH2COONH4(s) ��H��0
NH2COONH4(s) ��H��0 
��1��ʵ�����Ʊ�NH3�Ļ�ѧ����ʽ�ǣ� ��
��2���Ʊ���������淋�װ������ͼ��ʾ���Ѱ����Ͷ�����̼ͨ�����Ȼ�̼�У����Ͻ����ϣ����ɵİ��������С�������������Ȼ�̼�С���������϶�ʱ��ֹͣ�Ʊ���

ע�����Ȼ�̼��Һ��ʯ����Ϊ���Խ��ʡ�
�ٷ������ñ�ˮ��ȴ��ԭ���� ��Һ��ʯ������ƿ�������� ��
�ڴӷ�Ӧ��Ļ�����з������Ʒ��ʵ�鷽���� (��д��������)��Ϊ�˵õ������Ʒ��Ӧ��ȡ�ķ����� (��дѡ�����)��
a. ��ѹ���Ⱥ�� b. ��ѹ���Ⱥ�� c. ���40 �����º��
��β������װ������ͼ��ʾ��˫ͨ�����ܵ����ã� ��
Ũ��������ã� �� �� ����������������
��3��ȡ�ֱ��ʶ�����̼����淋İ����������Ʒ0.7825 g��������ʯ��ˮ��ִ�����ʹ̼Ԫ����ȫת��Ϊ̼��ƣ����ˡ�ϴ�ӡ�����������Ϊ1.000 g������Ʒ�а�������淋����ʵ�������Ϊ ��
��1��2NH4Cl + Ca(OH)2��2NH3 ��+ CaCl2 + 2H2O(δд������1��)(2��)
��2���� �����¶ȣ���߷�Ӧ��ת����(���¶ȣ���ֹ��Ӧ������ɲ���ֽ�)(2��)��ͨ���۲����ݣ�����NH3��CO2ͨ�����(2��)�� ����(1��) c(1��)
�۷�ֹ����(2��)���ն��ఱ������ֹ������ˮ�������뷴Ӧ��ʹ���������ˮ��(2��)
��3��75%(3/4��0.75���������������)(3��)
��2���� �����¶ȣ���߷�Ӧ��ת����(���¶ȣ���ֹ��Ӧ������ɲ���ֽ�)(2��)��ͨ���۲����ݣ�����NH3��CO2ͨ�����(2��)�� ����(1��) c(1��)
�۷�ֹ����(2��)���ն��ఱ������ֹ������ˮ�������뷴Ӧ��ʹ���������ˮ��(2��)
��3��75%(3/4��0.75���������������)(3��)
�����������1��ʵ�����Ʊ�NH3�Ļ�ѧ����ʽ��2NH4Cl + Ca(OH)2
 2NH3 ��+ CaCl2 + 2H2O��
2NH3 ��+ CaCl2 + 2H2O����2������Ϊ��Ӧ���ȣ������¶ȣ���߷�Ӧ��ת���ʣ�ͬʱ����ֹ��Ӧ������ɲ���ֽ⣻ͨ���۲����ݣ�����NH3��CO2ͨ������ڷ�Ӧ��Ĺ����Һ��Ļ����������Ʒ��ʵ�鷽���ǹ��ˡ���Ϊ������������ȶ��ԲΪ�˵õ������Ʒ��Ӧ��ȡ�ķ��������40 �����º�ɣ���ͼ��β������װ��ʹ���˽�����ϴ�IJ����ܣ��ɷ�ֹ������Ũ����������ն��ఱ������ֹ������ˮ�������뷴Ӧ��ʹ���������ˮ�⡣
��3����NH2COONH4����CaCO3��֪0.7825 g NH2COONH4��0.0133mol��1.000 g CaCO3��0.01mol����Ʒ�а�������淋����ʵ�������Ϊ75%��

��ϰ��ϵ�д�
 ���ſ����ϵ�д�
���ſ����ϵ�д� ���Ŀ����ϵ�д�
���Ŀ����ϵ�д� ������ӱ������ͯ������ϵ�д�
������ӱ������ͯ������ϵ�д�
�����Ŀ
 g
g
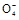 +2H2O+5SO2
+2H2O+5SO2 2Mn2++5S
2Mn2++5S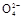 +4H+
+4H+