��Ŀ����
18��������������ȷ���ǣ��������ٱ�״���£�1L HCl��1L H2O�����ʵ�����ͬ��
�ڱ�״���£�11.2L H2��14g N2����ԭ������ͬ��
��28g CO�����ԼΪ22.4L��
���������ʵ����ʵ�����ͬ���������ڱ�״���µ������ͬ��
��ͬ��ͬ���ʱ���������ʵ����ʵ���Խ����ѹǿԽ��
��ͬ��ͬѹ�£�������ܶ����������Է������������ȣ�
| A�� | ���ݢ� | B�� | ���ڢݢ� | C�� | ���ڢۢݢ� | D�� | ���٢ڢܢݢ� |
���� �ٱ�״���£�H2OΪҺ�壻
�ڸ���n=$\frac{V}{{V}_{m}}$=$\frac{m}{M}$���㣻
��������ڵ����������δ֪��
�ܲ�һ��Ϊ���壻
�ݸ���P=nRT�жϣ�
���ݦ�=$\frac{m}{V}$=$\frac{nM}{n{V}_{m}}$=$\frac{M}{{V}_{m}}$�жϣ�
��� �⣺�ٱ�״���£�H2OΪҺ�壬��1LHCl�����ʵ�����ͬ���ʴ���
��11.2L H2�����ʵ���Ϊ0.5mol��14g N2�����ʵ���ҲΪ0.5mo�����ߺ�ԭ������ͬ������ȷ��
��������ڵ����������δ֪���ʴ���
�ܲ�һ��Ϊ���壬�ʴ���
����P=nRT��֪��ͬ��ͬ���ʱ���������ʵ����ʵ���Խ����ѹǿԽ����ȷ��
���ɦ�=$\frac{m}{V}$=$\frac{nM}{n{V}_{m}}$=$\frac{M}{{V}_{m}}$��֪��ͬ��ͬѹ�£�������ܶ����������Է������������ȣ�����ȷ��
��ѡB��
���� ���⿼���й����ʵ����ļ��㼰����٤�����ɼ������ۣ���ȷ��������������ʵ����Ĺ�ϵ����ȷ���ʵ�״̬��������״������ȷpV=nRT��pM=��RT���ɽ��

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
9���ڷ�Ӧ��11P+15CuSO4+24H2O=5Cu3P+6H3PO4+15H2SO4�У�15molCuSO4��������ԭ�ӵ����ʵ���Ϊ��������
| A�� | $\frac{1}{5}$mol | B�� | 2mol | C�� | 3 mol | D�� | 6mol |
6���й���������������ȷ���ǣ�������
| A�� | ������ζʱҪС�Ľ�����ƿ���ڱǿ���ֱ���� | |
| B�� | ��������Ư���ԣ�������ʹ�ʻ���ɫ | |
| C�� | ��ͨ������£��������Ժ�����ͭ�Ƚ���ֱ�ӻ��� | |
| D�� | ����й©ʱ����պ��̼������Һ��ë�����ڱǿ���Χ |
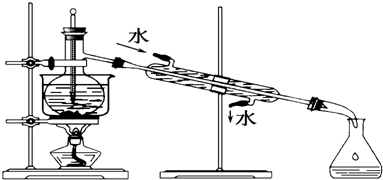
13��ֻ�ý�ͷ�ιܺ��Թ�ͨ�����Լ��Ļ��Σ��������������Һ��������
| A�� | NaAlO2��Al2��SO4��3 | B�� | NaHCO3��Ca��OH��2 | C�� | AlCl3�Ͱ�ˮ | D�� | HCl��Na2CO3 |