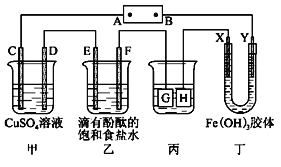
��Ŀ����
����Ŀ������˵����ȷ���ǣ�������
A. ��101kPaʱ��1mol H2��ȫȼ������Һ̬ˮ���ų�285.8kJ������H2��ȼ����Ϊ��285.8kJ��mol��1
B. �ⶨHCl��NaOH��Ӧ���к���ʱ��ÿ��ʵ���Ӧ����3���¶ȣ���������ʼ�¶ȣ�NaOH��ʼ�¶Ⱥͷ�Ӧ����ֹ�¶�
C. ��101k Paʱ��1mol C������O2��Ӧ����1mol COʱ���ų�110.5kJ��������C��ȼ����Ϊ110.5kJ��mol��1
D. ��ϡ��Һ�У�H��(aq)��OH��(aq)��H2O(l)��![]() H����57.3 kJ��mol��1��������0.5mol H2SO4��Ũ�����뺬1mol NaOH����Һ��ϣ��ų�����������57.3kJ
H����57.3 kJ��mol��1��������0.5mol H2SO4��Ũ�����뺬1mol NaOH����Һ��ϣ��ų�����������57.3kJ
���𰸡�B
��������
A��ȼ������ָ��һ�������£�1mol��ȼ����ȫȼ�������ȶ���������ʱ���ų���������A��û��ָ���¶ȣ���A����
B���к��Ȳⶨʱ������ⶨ��ʼ�¶Ⱥͷ�Ӧ���¶ȣ�����ÿ��ʵ���Ӧ����3���¶ȣ���������ʼ�¶ȣ�NaOH��ʼ�¶Ⱥͷ�Ӧ����ֹ�¶ȣ���B��ȷ��
C��̼�����ȶ�������Ϊ������̼������һ����̼�����Բ��ܱ�ʾ̼��ȼ���ȣ���C����
D����ΪŨ����ϡ�ͷ��ȣ����Խ���0.5molH2SO4��Ũ�����뺬1molNaOH����Һ��ϣ��ų�����������57.3kJ����D����
��ѡB��

����Ŀ����25��ʱ���������ʵĵ���ƽ�ⳣ�������ʾ��
��ѧʽ | CH3COOH | H2CO3 | HClO |
����ƽ�ⳣ�� | 1.7��10-5 | K1��4.3��10-7 K2��5.6��10-11` | 3.0��10-8 |
��ش��������⣺
��1���г�CH3COONa��ˮ��ƽ�ⳣ��Kh�ļ���ʽ��_________��
��2�����ʵ���Ũ����ͬ��������Һ��a.CH3COONa b��NaHCO3 c��NaClO
������Һ��pH��С�������е�˳����___<___< ____(�ñ����д)��
��3��д����NaClO��Һ��ͨ������CO2�Ļ�ѧ����ʽ________��
��.��4��̼������Һ�ʼ��Ե�ԭ���ǣ�_____________����д�����ӷ���ʽ����0.01mol/L Na2CO3��Һ��pH___0.1mol/LNa2CO3��Һ��pH������������������������=����
��5��Ũ�Ⱦ�Ϊ0.1mol/L�Ģ�CH3COONa��Һ��NH4Cl��Һ��ϡHCl��Na2SO4��Һ��pH�ɴ�С������˳��Ϊ_______��