��Ŀ����
Fe(OH)3����������������ҪӦ�ã�����FeCl3�ͷ�ˮ��Ӧ�Ʊ���Fe(OH)3�����г�����FeCl3��HCl����֪���岻������Ĥ����С���Ӻ����ӿ�������Ĥ���Իش������й����⣺
(1)ʵ������ȡFe(OH)3����ķ�����________����________������ȥ�����еĻ��������________����֤�������Ѿ��Ƴɡ�
(2)������Һ�д���Fe3����H���ķ�����__________________________________________
(3)��ȥ�����л��е�FeCl3��HCl�ķ����ǣ�_________________________________��
(4)�����ʵ��ķ���֤�������Cl���Ѿ����룿___________________________��
(5)����Fe(OH)3�����FeCl3��Һ��ķ�����_________________________________��
(6)��������(Na2FeO4)�������Ա�KMnO4��ǿ������һ�ֱ��ܹ�ע�����;�ˮ������ָ��Na2FeO4��ˮ��ԭ������˵���þ�ˮ���к���Խ��_______________________________________��
(1)�ڷ��ڵ�����ˮ�еμӱ���FeCl3��Һ������Һ�ʺ��ɫʱ��ֹͣ���ȣ����Ƶý��塡���ˡ������
(2)ȡ������Һ���μ�KSCN��Һ�����ɫ˵����Fe3������ȡ������Һ���μ���ɫʯ����Һ�����ɫ˵����H��
(3)������װ���Ĥ�У�Ȼ����������ˮ��(����)
(4)ȡ��Ĥ�����һ�ε���Һ�������Թ��У����������ữ��AgNO3��Һ��������������֤�������Ѿ�����
(5)�۲���ɫ
(6)Na2FeO4�к�ǿ�������ԣ�����Ч��ɱ��ˮ�е�ϸ���Ͳ������仹ԭ����Fe3���ܷ���ˮ�ⷴӦ����Fe(OH)3���壬��������ˮ�е����ʡ�Na2FeO4�����������;��������У��������κζ������к�������
����

 �Ķ��쳵ϵ�д�
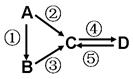
�Ķ��쳵ϵ�д�������ͨ��������Һʱ���Ӳ���۲쵽һ�������ġ�ͨ·����˵��������Һ��
| A������ | B������Һ | C����Һ | D������Һ |
��1��ֻ��һ���Լ����ɳ�ȥ�������ʺͼ������ʡ��������ڿո�
| ��� | ���� | �Լ����ƻ�ѧʽ |
| �� | �����ʣ�NaHCO3��Һ��Na2CO3�� | |
| �� | �����ʣ�SiO2��CaCO3�� | |
| �� | �����ʣ�FeCl2��Һ��FeCl3�� | |
| �� | ����Na2CO3 Na2SiO3 Na2SO3��Һ | |
| �� | ���𣺣�NH4��2SO4 NH4C1 Na2SO4��Һ | |
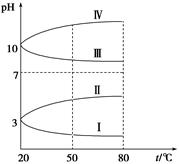
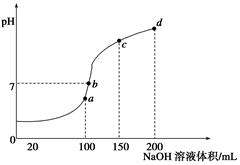
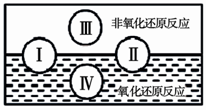
��2��������ԭ�dz����ķ�Ӧ���ͣ�������Ϣ�ش��������⣺ �����ֻ�����Ӧ������������ԭ��Ӧ�Ĺ�ϵ��Ҳ������ͼ����� �Т�Ϊ���Ϸ�Ӧ�����Ϊ�� ��Ӧ��д��һ����ˮ���ɵķ��Ϸ�Ӧ���͢�Ļ�ѧ����ʽ�� ��д��һ����ˮ�μӵķ��Ϸ�Ӧ���͢��Ļ�ѧ����ʽ ������ˮ��Ϊ ������������������ԭ���ȣ�
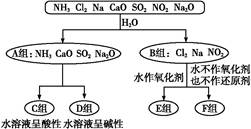
��3����SO2��Cl2��1��1����ȣ�ͬ��ͬѹ����Ϻ�ͨ��Ʒ����Һ�У���Һ����ɫ����ʵ��֤����SO2��Cl2����Һ�з�����������ԭ��Ӧ����SO2���������÷�Ӧ�����ӷ���ʽ�� ��
�������̽��Ҫ�õ����ѧ֪ʶ��
(1)�±��г������������Ħ���������ڱ�����д����Ħ�����������������
| ���� | �������ͯ���� | �������������� | �л������� |
| Ħ���� | �������� | ̼��� | �������� |
| Ħ�������������(ָ�ᡢ��Ρ������������������) | | | |
(2)��������Ʋ⣬����Ħ�������ܽ�����________________________
(����ܡ������ܡ�)��
(3)�����е�Ħ����̼��ƿ�����ʯ��ʯ���Ʊ���ijѧ�������һ��ʵ�����Ʊ�̼��Ƶ�ʵ�鷽����������ͼΪ��

��д�������������йط�Ӧ�Ļ�ѧ����ʽ����ע����Ӧ���ͣ�
��________________________________________________��
��________________________________________________��
��________________________________________________��
(4)��������ʯ��ʯ��ԭ��(�����Լ���ѡ)�����ʵ�����Ʊ�̼��Ƶ���һ��ʵ�鷽��������(3)��ʾ�������ʵ�鷽��������ͼ��ʾ������
ʯ��ʯ�D��
����Ƶķ������ŵ�Ϊ��________________________________��
(5)�����������Ƿ���̼��Ƶ�ʵ�鷽���ǣ�__________________��
���з�Ӧ�����ӷ���ʽ��ȷ����
| A��̼�����ϡ���ᷴӦ�� CaCO3+2H+�� Ca2++ CO2��+H2O |
| B����������ˮ��Ӧ�� 2Na +2H2O ��2Na+ +2OH��+ H2�� |
| C��������ϡ�����У� 2Fe��6H+�� 2Fe3+��3H2�� |
| D������������Һ�еμ�ϡ���Ba2+ + SO42����BaSO4�� |