ƒøƒ⁄»ð
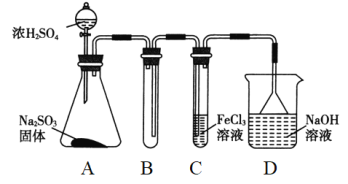
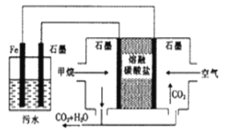
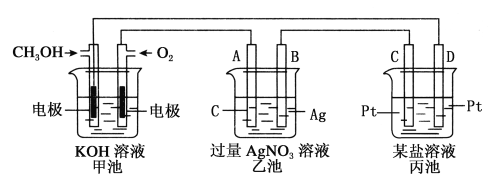
°æƒø°ø¡Úº∞∆‰ªØ∫œŒÔ”––Ì∂ý”√Õæ°£«Îªÿ¥œ¬¡–Œ °£
![]() ª˘Ã¨¡Ú¿Î◊”º€≤„µÁ◊”µƒπϵ¿±Ì¥Ô ΩŒ™____£¨∆‰µÁ◊”ÃÓ≥‰µƒ◊Ó∏þƒÐº∂µƒπϵ¿ ˝Œ™____°£
ª˘Ã¨¡Ú¿Î◊”º€≤„µÁ◊”µƒπϵ¿±Ì¥Ô ΩŒ™____£¨∆‰µÁ◊”ÃÓ≥‰µƒ◊Ó∏þƒÐº∂µƒπϵ¿ ˝Œ™____°£
![]() ≥£º˚∫¨¡ÚµƒŒÔ÷ ”–µ•÷ ¡Ú
≥£º˚∫¨¡ÚµƒŒÔ÷ ”–µ•÷ ¡Ú![]() °¢
°¢![]() °¢
°¢![]() °¢
°¢![]() µ»£¨Àƒ÷÷ŒÔ÷ µƒ»€µ„”…∏þµΩµÕµƒÀ≥–Ú“¿¥ŒŒ™__________°£
µ»£¨Àƒ÷÷ŒÔ÷ µƒ»€µ„”…∏þµΩµÕµƒÀ≥–Ú“¿¥ŒŒ™__________°£
![]() ∑Ω«¶øÛ
∑Ω«¶øÛ![]() º¥¡ÚªØ«¶
º¥¡ÚªØ«¶![]() «“ª÷÷±»Ωœ≥£º˚µƒøیԣ¨À·»Ð∑¥”¶Œ™£∫
«“ª÷÷±»Ωœ≥£º˚µƒøیԣ¨À·»Ð∑¥”¶Œ™£∫![]() ≈®
≈®![]() £¨‘Ú
£¨‘Ú![]() ÷–≈‰Œª‘≠◊” «__________£¨µ⁄“ªµÁ¿ÎƒÐ
÷–≈‰Œª‘≠◊” «__________£¨µ⁄“ªµÁ¿ÎƒÐ![]() __________
__________![]() ÃÓ°∞
ÃÓ°∞![]() °±°¢°∞
°±°¢°∞![]() °±ªÚ°∞
°±ªÚ°∞![]() °±
°±![]() £¨
£¨![]() ÷–¡Úµƒ‘”ªØ∑Ω ΩŒ™______________£¨œ¬¡–∆¯Ã¨∑÷◊”µƒø’º‰ππ–Õ”Î
÷–¡Úµƒ‘”ªØ∑Ω ΩŒ™______________£¨œ¬¡–∆¯Ã¨∑÷◊”µƒø’º‰ππ–Õ”Î![]() œýÕ¨µƒ”–__________°£
œýÕ¨µƒ”–__________°£
A.SnCl2 B.SO3 ![]()
![]()
![]() ∑Ω«¶øÛµƒ¡¢∑Ωæß∞˚»ÁÕºÀ˘ 棨¡Ú¿Î◊”≤…»°√Ê–ƒ¡¢∑Ω∂—ª˝£¨«¶¿Î◊”ÃÓ‘⁄”…¡Ú¿Î◊”–Œ≥…µƒ__________ø’œ∂÷–°£“—÷™æßÃÂ√Ð∂»Œ™¶—g°§cm£≠3£¨∞¢∑¸º”µ¬¬Þ≥£ ˝µƒ÷µŒ™
∑Ω«¶øÛµƒ¡¢∑Ωæß∞˚»ÁÕºÀ˘ 棨¡Ú¿Î◊”≤…»°√Ê–ƒ¡¢∑Ω∂—ª˝£¨«¶¿Î◊”ÃÓ‘⁄”…¡Ú¿Î◊”–Œ≥…µƒ__________ø’œ∂÷–°£“—÷™æßÃÂ√Ð∂»Œ™¶—g°§cm£≠3£¨∞¢∑¸º”µ¬¬Þ≥£ ˝µƒ÷µŒ™![]() £¨‘Úæß∞˚÷–¡Ú¿Î◊””Ϋ¶¿Î◊”◊ÓΩ¸µƒæý¿ÎŒ™__________nm°£
£¨‘Úæß∞˚÷–¡Ú¿Î◊””Ϋ¶¿Î◊”◊ÓΩ¸µƒæý¿ÎŒ™__________nm°£

°æ¥∞∏°ø![]() 3
3 ![]()
![]()
![]()
![]() AC Àƒ√ÊÃÂ
AC Àƒ√Êà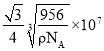
°æΩ‚Œˆ°ø
¢≈ª˘Ã¨¡Ú‘≠◊”µÁ◊”≈≈≤º Ω1s22s22p63s23p4£¨¿Î◊”µÁ◊”≈≈≤º Ω1s22s22p63s23p6£¨¿Î◊”º€≤„µÁ◊”≈≈≤º ΩŒ™3s23p6£¨¿Î◊”º€µÁ◊”µƒπϵ¿±Ì¥Ô ΩŒ™![]() £¨
£¨
π ¥∞∏Œ™![]()
∆‰µÁ◊”ÃÓ≥‰µƒ◊Ó∏þƒÐº∂Œ™3pƒÐº∂£¨3pƒÐº∂µƒπϵ¿ ˝Œ™3∏ˆπϵ¿£¨
π ¥∞∏Œ™3£ª
¢∆ŒÔ÷ µƒ»€µ„±»Ωœµƒ“ª∞„‘≠‘ÚŒ™¿Î◊”æßû€µ„¥Û”⁄∑÷◊”æßû€µ„£¨¡ÚªØƒ∆∫աڪغÿ¡Ω’þ∂ºŒ™¿Î◊”æßã¨∏˘æð¿Î◊”∞Îæ∂∫Õæß∏҃б»Ωœ£¨ƒ∆¿Î◊”∞Îæ∂–°”⁄ºÿ¿Î◊”∞Îæ∂£¨¡ÚªØƒ∆æß∏҃Х۔⁄¡ÚªØºÿæß∏҃У¨“Ú¥À¡ÚªØƒ∆»€µ„¥Û”⁄¡ÚªØºÿ»€µ„£¨∂˛—ıªØ¡Ú∫ÕS8∂º «∑÷◊”æßã¨◊È≥…Ω·ππœýÀ∆µƒ∑÷◊”æß㨜ý∂‘∑÷◊”÷ ¡ø‘Ω¥Û»€µ„‘Ω∏þ£¨“Ú¥ÀS8»€µ„¥Û”⁄∂˛—ıªØ¡Ú»€µ„£¨À˘“‘Àƒ’þ»€µ„∏þµÕÀ≥–ÚŒ™Na2S > K2S > S8 > SO2£¨
π ¥∞∏Œ™Na2S > K2S > S8 > SO2
¢«‘ÚH2[PbCl4]÷–≈‰Œª‘≠◊” «Cl£¨π ¥∞∏Œ™Cl£ª
Õ¨÷Ð∆⁄µ⁄“ªµÁ¿ÎƒÐ¥”◊ÛµΩ”“‘ˆ¥Û£¨µ´µ⁄IIA>III A,V A> VI A£¨¬»µƒµ⁄“ªµÁ¿ÎƒÐ¥Û”⁄¡Úµƒµ⁄“ªµÁ¿ÎƒÐ£¨π ¥∞∏Œ™![]() £ª
£ª
H2S÷–µÁ◊”∂‘ ˝
![]()
À˘“‘H2Sµƒ‘”ªØ∑Ω ΩŒ™SP3£¨
π ¥∞∏Œ™SP3£¨
H2S÷–µÁ◊”∂‘ ˝=2£´2=4£¨VSEPRƒ£–ÕŒ™’˝Àƒ√Ê㨬‘»•¡Ω∂‘π¬∂‘µÁ◊”£¨Œ™°∞V°±–ŒΩ·ππ£¨SnCl2÷–µÁ◊” ˝=2£´2=4£¨Õ¨—˘Œ™°∞V°±–ŒΩ·ππ£¨SO3÷–µÁ◊” ˝=3£´0=3£¨º¥Œ™∆Ω√Ê»˝Ω«–ŒΩ·ππ£¨O3÷–¶ƒŒ™2£¨”…”⁄–Œ≥…¡À![]() £¨÷––ƒ—ı‘≠◊”
£¨÷––ƒ—ı‘≠◊”![]() –Œ≥… ±”√¡À¡Ω∏ˆµÁ◊”£¨“Ú¥Àªπ £”ý¡Ω∏ˆµÁ◊”º¥“ª∂‘π¬∂‘µÁ◊”£¨µÁ◊”∂‘ ˝=2£´1=3£¨º¥Œ™°∞V°±–ŒΩ·ππ£¨CH4÷–µÁ◊” ˝=4£´0=4£¨º¥Œ™’˝Àƒ√ÊÃÂΩ·ππ£ª
–Œ≥… ±”√¡À¡Ω∏ˆµÁ◊”£¨“Ú¥Àªπ £”ý¡Ω∏ˆµÁ◊”º¥“ª∂‘π¬∂‘µÁ◊”£¨µÁ◊”∂‘ ˝=2£´1=3£¨º¥Œ™°∞V°±–ŒΩ·ππ£¨CH4÷–µÁ◊” ˝=4£´0=4£¨º¥Œ™’˝Àƒ√ÊÃÂΩ·ππ£ª
π ¥∞∏Œ™AC
¢»∏˘æð∑Ω«¶øÛµƒ¡¢∑Ωæß∞˚£¨¡Ú¿Î◊”≤…»°√Ê–ƒ¡¢∑Ω∂—ª˝£¨Àƒ∏ˆ¡Ú¿Î◊”∫Õ«¶¿Î◊”–Œ≥…¡ÀÀƒ√ÊÃÂΩ·π𣨫¶¿Î◊”‘⁄Àƒ√ÊÃÂø’œ∂÷–£¨π ¥∞∏Œ™Àƒ√Êãª
“—÷™æßÃÂ√Ð∂»Œ™¶—g°§cm£≠3£¨
‘Úæß∞˚÷–¡Ú¿Î◊””Ϋ¶¿Î◊”◊ÓΩ¸µƒæý¿ÎŒ™ÃÂ∂‘Ω«œþµƒÀƒ∑÷÷Æ“ª£¨∏√æßÃÂ÷–”–4∏ˆ«¶¿Î◊”∫Õ4∏ˆ¡Ú¿Î◊”£¨…Ë¿‚≥§Œ™a£¨
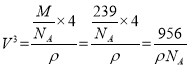
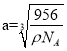
![]()
º¥æß∞˚÷–¡Ú¿Î◊””Ϋ¶¿Î◊”◊ÓΩ¸µƒæý¿ÎŒ™
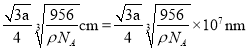
π ¥∞∏Œ™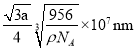

 ÃÏÃϜڅœ“ª±æ∫√æÌœµ¡–¥∞∏
ÃÏÃϜڅœ“ª±æ∫√æÌœµ¡–¥∞∏ –°—ß…˙10∑÷÷””¶”√Âœµ¡–¥∞∏
–°—ß…˙10∑÷÷””¶”√Âœµ¡–¥∞∏°æƒø°øµ™µƒµ•÷ º∞“ª–©ªØ∫œŒÔ‘⁄π§≈©“µ…˙≤˙µ»¡Ï”Ú”–÷ÿ“™”¶”√£¨ªÿ¥œ¬¡–Œ Â
(1)Ù«∞∑ (  ) «≤ªŒ»∂®µƒ∞◊…´¥Û∆¨◊¥ªÚ’Î◊¥Ω·æß°£º´“◊Œ¸≥±£¨º´“◊»Ð”⁄ÀÆ°£
) «≤ªŒ»∂®µƒ∞◊…´¥Û∆¨◊¥ªÚ’Î◊¥Ω·æß°£º´“◊Œ¸≥±£¨º´“◊»Ð”⁄ÀÆ°£
¢ŸÙ«∞∑∑÷◊”÷–≤…”√sp3‘”ªØµƒ‘≠◊” «____________________£ªÙ«∞∑º´“◊»Ð”⁄ÀÆ£¨÷˜“™‘≠“Ú «________________________________°£
¢⁄‘⁄Ù«∞∑µƒ◊È≥…µƒ‘™Àÿ÷–£¨¥¶”⁄Õ¨“ª÷Ð∆⁄‘™Àÿµƒµ⁄ÀƒµÁ¿ÎƒÐ(I4)Ωœ¥Ûµƒ «______________(”√‘™Àÿ∑˚∫≈±Ì æ)°£
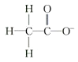
(2) þ¡ý§![]() ∫Õ¡⁄∑»¬Þþ¯∂º «∫¨µ™µƒ”–ª˙ŒÔ£¨l molþ¡ý§÷–∫¨”–
∫Õ¡⁄∑»¬Þþ¯∂º «∫¨µ™µƒ”–ª˙ŒÔ£¨l molþ¡ý§÷–∫¨”–![]() º¸µƒŒÔ÷ µƒ¡øŒ™_________ mol°£
º¸µƒŒÔ÷ µƒ¡øŒ™_________ mol°£
(3)“—÷™£∫√ø1molœ¬¡–ŒÔ÷ ∑÷Ω‚Œ™∆¯Ã¨ª˘Ã¨‘≠◊”œ˚∫ƒƒÐ¡ø∑÷±Œ™
NO2 | CO | CO2 | NO |
812kJ | 1076kJ | 1490kJ | 632kJ |
¢ŸNO2 + CO ![]() CO2 + NO
CO2 + NO
¢⁄N2(g)+O2(g) ![]() 2NO(g) ¶§H£Ω+179.5 kJ/mol
2NO(g) ¶§H£Ω+179.5 kJ/mol
¢€2NO(g) +O2(g)![]() 2NO2(g) ¶§H£Ω-112.3 kJ/mol
2NO2(g) ¶§H£Ω-112.3 kJ/mol
‘–¥≥ˆNO”ÎCO∑¥”¶…˙≥…ŒÞŒ€»æŒÔ∆¯Ãµƒ»»ªØ—ß∑Ω≥à Ω_________________°£