��Ŀ����
����Ŀ��ij���dz��Ը���Ϊԭ�����ǣ�ͬʱ�õ������ĸ��������Ը����������ۺ����ò�����������ۺ�Ч�棬���һ��ܷ�ֹ������Ⱦ������������ͼ��

��֪:ʯ���ѽ��ѳ�Ϊ����H����Ҫ������G�Ǿ�����ζ��Һ�塣�ش��������⣺
(1)H�еĹ���������Ϊ____________;E������Ϊ____________��
(2)H��D�Ļ�ѧ��Ӧ����Ϊ____________��
(3)ʵ�������Ʊ�G�ķ�Ӧװ����ͼ��ʾ��

����̲��в��õ�ʵ��װ�ò�ͬ����װ���в��������θ���ܣ���������____________������̼������Һ������____________��
��ʵ���������Ҫ�����Թ�a�е�G��Ҫ�õ�����Ҫ����������____________���ձ���
����ʵ�����ú�18O��D��F��Ӧ��F��G�Ļ�ѧ����ʽΪ____________��
���𰸡�̼̼˫�� ��ȩ �ӳɷ�Ӧ ������ �к����ᣬ�����Ҵ������������������ܽ�� ��Һ©�� CH3COOH��CH3CH218OH![]() CH3CO18OCH2CH3��H2O
CH3CO18OCH2CH3��H2O
��������
����������֮��õ���ά�أ���ά��ˮ������ղ���Ϊ�����ǣ�����A����ά�أ�B�������ǣ��������ھƻ�ø�������·�Ӧ�����Ҵ�����D���Ҵ���D��������������EΪCH3CHO��E��һ������������Ӧ����FΪCH3COOH��F��D����������Ӧ����GΪCH3COOCH2CH3��H��ˮ�ӳɵ��Ҵ�����HΪCH2=CH2��
(1)���ݷ�����֪��H����ϩ���ṹʽΪCH2=CH2������������Ϊ̼̼˫����E������Ϊ��ȩ��
(2)H��D����ϩ��ˮ�����ӳɷ�Ӧ�����Ҵ�����ѧ��Ӧ���ͼӳɷ�Ӧ��
(3)����̲��в��õ�ʵ��װ�ò�ͬ����װ���в��������θ���ܣ��������Ƿ�ֹ�������Ҵ������ᶼ���лӷ��ԣ����������л����ٲ����Ҵ������ᣬ�ڱ���̼������Һ�У������������ܽ�Ƚ�С���ʱ���̼������Һ�������к����ᣬ�����Ҵ������������������ܽ�ȣ�
��ʵ���������Ҫ�����Թ�a�е�G��GΪCH3COOCH2CH3�����������ͱ���̼������Һ�������ܣ����Բ��÷�Һ�ķ������룬��Һ��Ҫ�õ�����Ҫ���������Ƿ�Һ©�����ձ���
��D���Ҵ���FΪCH3COOH��������Ҵ�����������Ӧʱ��������ȥ�ǻ�������ȥ�ǻ��ϵ���ԭ�ӣ��ú�18O��D��F��Ӧ��F��G�Ļ�ѧ����ʽΪ��CH3COOH��CH3CH218OH![]() CH3CO18OCH2CH3��H2O��
CH3CO18OCH2CH3��H2O��

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�����Ŀ��ij�о���ѧϰС���������̿�(��Ҫ�ɷ���MnO2)���չ�ҵ�����е�SO2�����Ʊ������̵�������������(����Һ��pH<2�����г���Mn2+�⣬����������Fe2+��Al3+��Ca2+�Ƚ�������)��
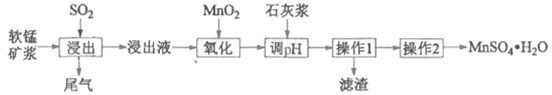
������1������Һͨ����������������ȥCa2+�����������γ������������ʱ��pH���±���
���� | ��ʼ����ʱ��pH | ��ȫ����ʱ��pH |
Fe2+ | 7.6 | 9.7 |
Fe3+ | 2.7 | 3.7 |
Al3+ | 3.8 | 4.7 |
Mn2+ | 8.3 | 9.8 |
(1)���������б����������ʵĻ�ѧʽΪ____________��
(2)������������Ҫ��Ӧ�����ӷ���ʽΪ_____________
(3)���������Һ���м���ʯ�ҽ�����pH�� pH �ķ�Χ��__________��
(4)����2��������Ũ����__________�� ���ˡ� ϴ�ӡ�����Ȳ�����
(5)Ϊ�ⶨ����MnSO4��H2O�Ĵ��ȣ�ȷ��ȡ������Ʒ1.720 g��������H2SO4��NH4NO3��Һ������ʹMn2+ȫ��������Mn3+�����200 mL��Һ��ȡ20.00 mL����Һ����������μ���0.0500 mol��L-1��FeSO4��Һ���������·�Ӧ�� Fe2+ +Mn3+=== Fe3+ + Mn2+����Ӧǡ����ȫ����ʱ��������FeSO4��Һ19.50 mL������MnSO4��H2O�Ĵ���(д��������̣����������λ��Ч����)__
����Ŀ��ʹ������к͵ζ����ⶨ���۰״���������g/100mL����
��ʵ�鲽�裺
��1������100mL����״���Һ����ȡ10.00mLʳ�ð״ף�ע���ձ�����ˮϡ�ͺ�ת�Ƶ�_____�����������ƣ��ж��ݣ�ҡ�ȼ��ã�
��2��ȡ����״���Һ20.00mL����ƿ�У������еμ�2��_____��ָʾ����
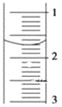
��3����ȡʢװ0.1000mol/L NaOH��Һ��______�����������ƣ��ij�ʼ���������Һ��λ������ͼ��ʾ�����ʱ�Ķ���Ϊ______mL��
��4���ζ�����__________ʱ��ֹͣ�ζ�������¼NaOH��Һ���ն������ظ��ζ�3�Ρ�
��ʵ���¼
1 | 2 | 3 | 4 | |
V����Ʒ�� | 20.00 | 20.00 | 20.00 | 20.00 |
V��NaOH�������ģ� | 15.95 | 15.00 | 15.05 | 14.95 |
�����ݴ��������ۣ�
��5�������㣬���۰״�������=_______g/100mL (�����ʽ��=60)��
��6���ڱ�ʵ������У����в�����ʹʵ����ƫ�����______����д��ţ�
A����ʽ�ζ����ڵζ�ʱδ�ñ�NaOH��Һ��ϴ
B����ʽ�ζ����ڼ����ڵζ�ǰ�����ݣ��ζ���������ʧ
C����ƿ�м������״���Һ���ټ�����ˮ
D����ƿ�ڵζ�����ҡ����������Һ�彦��