��Ŀ����
�������ڹ�ҵ�Ͼ��й㷺��Ӧ�á�
I���ݱ�����һ������������FeO2����鷴Ӧ����ȡ���������Ľ��������䷴Ӧ���£�
Fe2O3(s)��3CH4(g)��2Fe(s)��3CO(g)��6H2(g) ��H��0
��1������Ӧ��2L���ܱ������н��У�5min��ﵽƽ�⣬��÷�Ӧ����Fe������Ϊl.12g����ö�ʱ����CH4��ƽ����Ӧ����Ϊ ________________��
��2����Ӧ�ﵽƽ����������������䣬���ı�ijһ���Ԫ�أ�����˵����ȷ����___(ѡ�����)��
a�������������v������ƽ�������ƶ�
b�����¶����ߣ�ƽ�ⳣ��K��С
c��������Fe2O3������ƽ�������ƶ�
d�����ӷ�Ӧ��ϵ�����߲���CO�������CH4��ת����
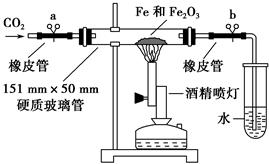
��ҵ����������������(��Ҫ�ɷ�ΪFe2O3��A12O3��SiO2��)Ϊԭ����ȡFe2O3������
�������£��Իش��������⣺

��3������i������A12O3�ܽ�����ӷ���ʽΪ________________��
��4������i����������Ҫ���еIJ���������__________________��
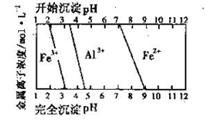
��5�������ͼ���жϲ��袢�е�����Һ��pH������_________________��
��6����֪

���㷴ӦFe2��(aq)��2HCO3��(aq) FeCO3(s)��H2CO3(aq)��ƽ�ⳣ��Ϊ_______��
FeCO3(s)��H2CO3(aq)��ƽ�ⳣ��Ϊ_______��
��7���ڿ���������FeCO3�����������Ļ�ѧ����ʽΪ____________��
��8�������ѧ��ѧ֪ʶ�����������ʵ���������������������������ȡ����������һ�ּ�����
I���ݱ�����һ������������FeO2����鷴Ӧ����ȡ���������Ľ��������䷴Ӧ���£�
Fe2O3(s)��3CH4(g)��2Fe(s)��3CO(g)��6H2(g) ��H��0
��1������Ӧ��2L���ܱ������н��У�5min��ﵽƽ�⣬��÷�Ӧ����Fe������Ϊl.12g����ö�ʱ����CH4��ƽ����Ӧ����Ϊ ________________��
��2����Ӧ�ﵽƽ����������������䣬���ı�ijһ���Ԫ�أ�����˵����ȷ����___(ѡ�����)��
a�������������v������ƽ�������ƶ�
b�����¶����ߣ�ƽ�ⳣ��K��С
c��������Fe2O3������ƽ�������ƶ�
d�����ӷ�Ӧ��ϵ�����߲���CO�������CH4��ת����
��ҵ����������������(��Ҫ�ɷ�ΪFe2O3��A12O3��SiO2��)Ϊԭ����ȡFe2O3������
�������£��Իش��������⣺

��3������i������A12O3�ܽ�����ӷ���ʽΪ________________��
��4������i����������Ҫ���еIJ���������__________________��
��5�������ͼ���жϲ��袢�е�����Һ��pH������_________________��

��6����֪

���㷴ӦFe2��(aq)��2HCO3��(aq)
 FeCO3(s)��H2CO3(aq)��ƽ�ⳣ��Ϊ_______��
FeCO3(s)��H2CO3(aq)��ƽ�ⳣ��Ϊ_______����7���ڿ���������FeCO3�����������Ļ�ѧ����ʽΪ____________��
��8�������ѧ��ѧ֪ʶ�����������ʵ���������������������������ȡ����������һ�ּ�����
��1��0.003mol/(L��min)û�е�λ��1�֣�
��2��d
��3��Al2O3��6H����2Al3����3H2O
��4������
��5��7
��6��4.06��106��4��1��106
��7��4FeCO3��O2 2Fe2O3��4CO2
2Fe2O3��4CO2
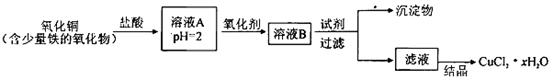
��8��ȡ�������������������������ȵ�Ũ����������Һ�����衢���ˡ�ϴ�ӡ���ɣ��𰸺������ɵ÷֣�
��2��d
��3��Al2O3��6H����2Al3����3H2O
��4������
��5��7
��6��4.06��106��4��1��106
��7��4FeCO3��O2
 2Fe2O3��4CO2
2Fe2O3��4CO2��8��ȡ�������������������������ȵ�Ũ����������Һ�����衢���ˡ�ϴ�ӡ���ɣ��𰸺������ɵ÷֣�
���������I����1��5min��ﵽƽ�⣬��÷�Ӧ����Fe������Ϊl.12g�����ʵ�����1.12g��56g/mol��0.02mol����˸��ݷ���ʽ��֪���ļ�������ʵ�����0.03mol����Ũ����0.015mol/L�������ü����ʾ�ķ�Ӧ������0.015mol/L��5min��0.003mol/(L��min)��
��2��a��������������淴Ӧ���ʾ�����ƽ�ⲻ�ƶ���a����ȷ��b������Ӧ�����ȷ�Ӧ��������¶����ߣ�ƽ��������Ӧ�����ƶ�������ƽ�ⳣ��K����b����ȷ��c���������ǹ��壬���������Fe2O3������ƽ�ⲻ�ƶ���c����ȷ��d�����ӷ�Ӧ��ϵ�����߲���CO������������Ũ�ȣ�ƽ��������Ӧ�����ƶ�����˿����CH4��ת���ʣ�d��ȷ����ѡd��
��3��A12O3�ܽ���ϡ�����з�Ӧ�����ӷ���ʽΪAl2O3��6H����2Al3����3H2O��
��4������i���������ж����������Թ��壬��˶�Ҫ���еIJ��������ǹ��ˡ�
��5������ͼ���֪���������ӿ�ʼ����ʱ��pHֵ��7������Ҫ�õ�̼������������Һ��pH���ܸ���7����˲��袢�е�����Һ��pH������7��
��6����ӦFe2��(aq)��2HCO3��(aq)
 FeCO3(s)��H2CO3(aq)��ƽ�ⳣ��K��
FeCO3(s)��H2CO3(aq)��ƽ�ⳣ��K��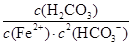 ��
��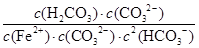 ��
��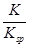 ��
��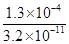 ��4.06��106��
��4.06��106����7������ԭ���غ��֪���ڿ���������FeCO3������������ͬʱ����CO2���ɣ���˷�Ӧ�Ļ�ѧ����ʽΪ4FeCO3��O2
 2Fe2O3��4CO2��
2Fe2O3��4CO2����8�����������������������ܺ�����������Һ��Ӧ����������������������Һ����Ӧ�����Ҫ��ȡ��������ʵ�鷽�������Ϊȡ�������������������������ȵ�Ũ����������Һ�����衢���ˡ�ϴ�ӡ���ɡ�

��ϰ��ϵ�д�
�����Ŀ
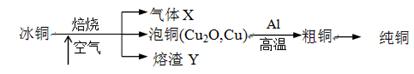

 3Zn��2K2FeO4��8H2O���õ�طŵ�ʱ�ĸ�����ӦʽΪ________�����ʱ����������Һ��pH________�����������䡱��С������
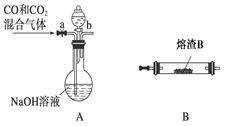
3Zn��2K2FeO4��8H2O���õ�طŵ�ʱ�ĸ�����ӦʽΪ________�����ʱ����������Һ��pH________�����������䡱��С������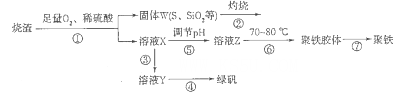
 2K2S+K2SO3+3H2O���÷�Ӧ���������뻹ԭ������֮��Ϊ___________��
2K2S+K2SO3+3H2O���÷�Ӧ���������뻹ԭ������֮��Ϊ___________��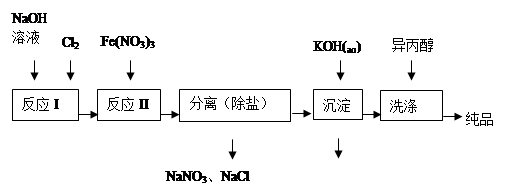
 6SO2��Fe3O4����3 mol FeS2�μӷ�Ӧ��ת��________ mol���ӡ�
6SO2��Fe3O4����3 mol FeS2�μӷ�Ӧ��ת��________ mol���ӡ�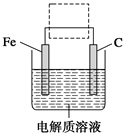
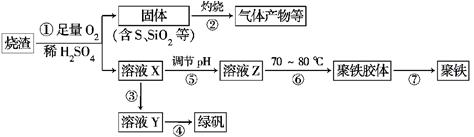
 xH2O)�������²�����
xH2O)�������²�����