��Ŀ����
��17�֣�H2O2��һ����ɫ������ԭ�Լ����ڻ�ѧ�о���Ӧ�ù㷺��
��1��ijС������ͬŨ��Fe3+�Ĵ��£�̽��H2O2Ũ�ȶ�H2O2�ֽⷴӦ���ʵ�Ӱ�졣��ѡ�Լ���������30% H2O2��0.1mol?L-1Fe2(SO4)3������ˮ����ƿ��˫������ˮ�ۡ����ܡ��������ܡ���Ͳ�����������ˮԡ�ۡ�ע����
��д����ʵ��H2O2�ֽⷴӦ����ʽ����������ת�Ƶķ������Ŀ��____________
�����ʵ�鷽�����ڲ�ͬH2O2Ũ���£��ⶨ ____________________________________��Ҫ������õ�������ֱ�����ַ�Ӧ���ʴ�С����
�����ʵ��װ�ã������ͼ��װ��ʾ��ͼ��

�ܲ����±���ʽ���ⶨʵ�������������ʵ�鷽�����г���ѡ�Լ���������¼�Ĵ��������������ⶨ�����ݣ���������ĸ��ʾ����

��2������ͼ21��a����21��b���е���Ϣ����ͼ21��c��װ�ã����ܵ�A��Bƿ���ѳ���NO2���壩����ʵ�顣�ɹ۲쵽Bƿ��������ɫ��Aƿ�е�_________������dz��������ԭ����______________________________________________________��

��1��ijС������ͬŨ��Fe3+�Ĵ��£�̽��H2O2Ũ�ȶ�H2O2�ֽⷴӦ���ʵ�Ӱ�졣��ѡ�Լ���������30% H2O2��0.1mol?L-1Fe2(SO4)3������ˮ����ƿ��˫������ˮ�ۡ����ܡ��������ܡ���Ͳ�����������ˮԡ�ۡ�ע����
��д����ʵ��H2O2�ֽⷴӦ����ʽ����������ת�Ƶķ������Ŀ��____________
�����ʵ�鷽�����ڲ�ͬH2O2Ũ���£��ⶨ ____________________________________��Ҫ������õ�������ֱ�����ַ�Ӧ���ʴ�С����
�����ʵ��װ�ã������ͼ��װ��ʾ��ͼ��

�ܲ����±���ʽ���ⶨʵ�������������ʵ�鷽�����г���ѡ�Լ���������¼�Ĵ��������������ⶨ�����ݣ���������ĸ��ʾ����

��2������ͼ21��a����21��b���е���Ϣ����ͼ21��c��װ�ã����ܵ�A��Bƿ���ѳ���NO2���壩����ʵ�顣�ɹ۲쵽Bƿ��������ɫ��Aƿ�е�_________������dz��������ԭ����______________________________________________________��

��1����
��������ͬ��������������ʱ��
��
�ܲⶨ��Ӧʱ��
��2�����Ϊ��������ֽ��Ƿ��ȷ�Ӧ��2NO2(g)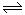 N2O4(g)Ҳ�Ƿ��ȷ�Ӧ������Bƿ�¶ȸ���Aƿ���¶����ߣ�ƽ�������ƶ�����������Ũ��������ɫ���
N2O4(g)Ҳ�Ƿ��ȷ�Ӧ������Bƿ�¶ȸ���Aƿ���¶����ߣ�ƽ�������ƶ�����������Ũ��������ɫ���

��������ͬ��������������ʱ��
��
�ܲⶨ��Ӧʱ��
| | H2O2�����(mL) | 0.1mol?L-1Fe2(SO4)3�������mL�� | ��������ˮ�������mL�� | ����O2�����(mL) | ��Ӧʱ�䣨min�� |
| ʵ��1 | b | a | c | d | |
| ʵ��2 | c | a | b | d | |
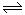 N2O4(g)Ҳ�Ƿ��ȷ�Ӧ������Bƿ�¶ȸ���Aƿ���¶����ߣ�ƽ�������ƶ�����������Ũ��������ɫ���
N2O4(g)Ҳ�Ƿ��ȷ�Ӧ������Bƿ�¶ȸ���Aƿ���¶����ߣ�ƽ�������ƶ�����������Ũ��������ɫ��������������1���ٹ��������������������������·ֽ�����ˮ�������������������������������ԭ������ѧ����ʽ������ת�Ʒ������ĿΪ

�ڷ�Ӧ�����ǵ�λʱ��������Ũ�ȵĸı��������Բⶨ��ͬŨ�ȵĹ�������Էֽ����ʵ�Ӱ�죬��ⶨ��ͬʱ���ڣ���������������Ķ��٣���������ͬ�������������ʱ��Ķ��٣�
��������ˮ���������ռ�һ�������O2����Ҫˮ�ۡ���Ͳ�������ܣ���Ͳ��ʢ��ˮ������ˮ���У�װ����ͼ
��
| | H2O2�����(mL) | 0.1mol?L-1Fe2(SO4)3�������mL�� | ��������ˮ�������mL�� | ����O2�����(mL) | ��Ӧʱ�䣨min�� |
| ʵ��1 | b | a | c | d | |
| ʵ��2 | c | a | b | d | |
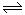 N2O4(g)������У���������Ũ������Bƿ����ɫ��Aƿ����ɫ�
N2O4(g)������У���������Ũ������Bƿ����ɫ��Aƿ����ɫ�
��ϰ��ϵ�д�
�����Ŀ
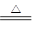 5NaCl+NaClO3+3H2O������������ȷ����
5NaCl+NaClO3+3H2O������������ȷ����
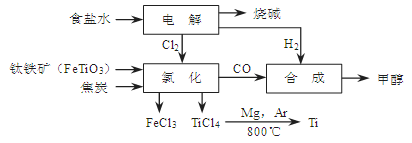
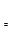 CH3OH(g)��
CH3OH(g)��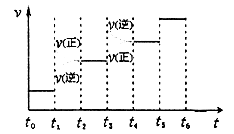


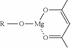 �����ɷ������·�Ӧ��
�����ɷ������·�Ӧ��