��Ŀ����
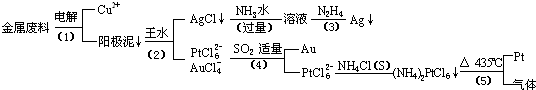
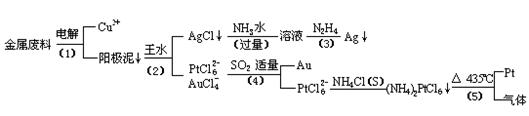
�Ӻ�����ͭ����Ͳ��Ľ�����������ȡ����������һ�ֹ������£�
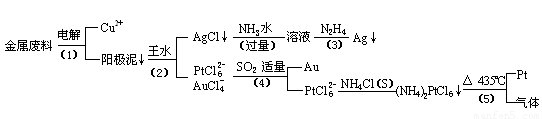
�������Ϲ��ջش��������⣺
��1�� ���ʱ���Խ�������Ϊ��������ͭΪ������CuSO4��ҺΪ���Һ��д����ⷽ��ʽ��
������
������ ��
��2��AgCl���ڰ�ˮ�����õ���Һ���е�һ��������,�ڼ��������£�Ҳ���������ǽ��仹ԭΪ����д���÷�Ӧ�����ӷ���ʽ��
��
��3��д�����裨4�������ӷ�Ӧ����ʽ��
��
��4�����Ũ���ᷴӦ�Ļ�ѧ����ʽΪ��
Au + 6HNO3��Ũ��= Au(NO3)3 + 3NO2��+ 3H2O
���÷�Ӧ��ƽ�ⳣ����С�����Խ��Ũ���Ἰ������Ӧ������ȴ����������ˮ���Լ�Ҫ����֮��
��
��1��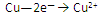 ��
��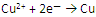 (��1��)
(��1��)
��2��2Ag(NH3)2+ + 2OH�� + CH2OH(CHOH)4CHO �� 2Ag + 4NH3 + CH2OH(CHOH)4COOH + H2O(3��)
��3��2AuCl4- + 3SO2 + 6 H2O = 2Au + 8Cl- + 3SO42- + 12H+ (3��)
��4����ˮ�к��д�����Cl����Au3+��Cl��������AuCl4����ʹ��ƽ����Au3+Ũ�Ƚ��ͣ�ƽ�����ƣ���������ˮ��(3��)
����������

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
 2Ag+4NH3+CH2OH��CHOH��4COOH+H2O
2Ag+4NH3+CH2OH��CHOH��4COOH+H2O