��Ŀ����
17��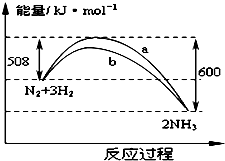 ��ͼ��425��ʱN2��H2��Ӧ�����������仯������ͼ������������ȷ���ǣ�������
��ͼ��425��ʱN2��H2��Ӧ�����������仯������ͼ������������ȷ���ǣ�������| A�� | �÷�Ӧ���Ȼ�ѧ����ʽΪ��N2+3H2?2NH3��H=-92kJ•mol-1 | |
| B�� | һ��������N2+3H2�T2NH3�ﵽƽ��ʱ��3v����H2��=2v����NH3�� | |
| C�� | ����������÷�Ӧ�ķ�Ӧ�ȷ����ı� | |
| D�� | �¶ȡ��������һ����ͨ��1molN2��3molH2��Ӧ��ų�������ΪQ1kJ����ͨ��2 molN2��6 molH2��Ӧ��ų�������ΪQ2kJ������184��Q2��2Q1 |
���� A�������Ȼ�ѧ����ʽ����дԭ���жϣ���H=�ų�������-���յ�������
B��N2+3H2�T2NH3�ﵽƽ��ʱ�����淴Ӧ������ȣ�
C�����ݴ������ܸı䷴Ӧ������������������������IJ�ֵ������
D���¶ȡ��������һ����ͨ��1molN2��3molH2��Ӧ����Ӧ���ܽ��г��ף���ͨ��2 molN2��6 molH2��Ӧ������㶨����Ӧ���������ʵ������ӣ���Ӧ�ȼӱ�������ѹǿ��ƽ���Ӱ�������
��� �⣺A���÷�Ӧ�ų��������������յ�����������Ϊ���ȷ�Ӧ���Ȼ�ѧ����ʽΪ��N2��g��+3H2��g��=2NH3��g����H=-92kJ•mol-1��Ҫ�������ʵľۼ�״̬����A����
B��һ��������N2+3H2�T2NH3�ﵽƽ��ʱ�����淴Ӧ������ȣ���2v����H2��=3v����NH3������B����
C�������ܸı䷴Ӧ��·�����ӿ췴Ӧ���ʣ����Dz��ܸı䷴Ӧ������������������������IJ�ֵ������Ӧ�Ȳ��䣬��C����
D���¶ȡ��������һ����ͨ��1molN2��3molH2��Ӧ����Ӧ���ܽ��г��ף���Ӧ��ų�������Q1kJ��92kJ������ѹǿ���ӣ�ƽ��������ƶ����ų���������ԭ���ķ�Ӧ�ȵ�2����Ҫ�࣬����184��Q2��2Q1����D��ȷ��
��ѡD��
���� ���⿼�����Ȼ�ѧ����ʽ�����������ʡ�ƽ�⼰������Ӱ�죬ѹǿ��ƽ���ƶ���Ӱ�죬�漰��֪ʶ��϶࣬�Ѷ�һ�㣮

| A�� | �DZ�״���£�1mol�κ���������������Ϊ22.4L | |
| B�� | ��״���£�11.2LH2O���еķ�����Ϊ0.5NA | |
| C�� | 25�棬1.01��105Pa��16gCH4�к��е�ԭ����Ϊ5NA | |
| D�� | 92g NO2��N2O4�Ļ���ﺬ�еķ�����Ϊ2NA |
| A�� | ��AlCl3��Һ���ɵõ��Ĺ�����AlCl3 | |
| B�� | ��¯ˮ���е�CaSO4���ñ���Na2CO3��Һ���ݣ�����ϡ�����ܽ��ȥ | |
| C�� | ���ȴ�ˮ��KW���pH��С�������� | |
| D�� | �ڱ���BaSO4��Һ�м�������Na2SO4���壬��Һ��c��Ba2+������ |
��1��A��B��Ϊ�����ڽ���Ԫ�أ������±����ݣ�д��B�Ļ�̬ԭ�ӵĺ�������Ų�ʽ1s22s22p63s2��
| �����ܣ�kJ/mol�� | I1 | I2 | I3 | I4 |
| A | 932 | 1821 | 15390 | 21771 |
| B | 738 | 1451 | 7733 | 10540 |
 ��
����3��ij�������ṹ��ͼ1��ʾ�������ں��е���������ACE������ţ���
A�������B�����Ӽ���C�����ۼ���D�������� E����λ��
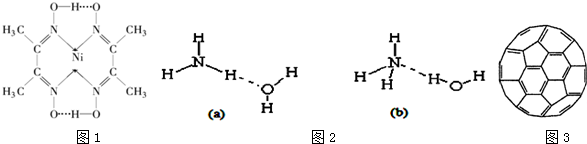
��4������NH3•H2O�ĵ��뷽��ʽΪNH3•H2O?NH4++OH-�����ж�NH3����ˮ���������NH3•H2O�Ľṹ������b ������� a �� b����ͼ2����
��5������ϩ��C60���Ľṹ��ͼ3��ʾ��1mol C60�����к���̼̼��������ĿΪ60NA��
| A�� | ˮ�����ɱ������ᡢ�ռ���� | B�� | ��ơ��������ᡢ�����ơ����� | ||
| C�� | �����������������ᡢ�������ͭ | D�� | ͭ������ͭ�����ᡢ��ʯ�ҡ�ʯ��ʯ |