��Ŀ����
 SO2�dz����Ĵ�����Ⱦ��֮һ���ҹ��涨������SO2�������ó���0.02mg/L��
SO2�dz����Ĵ�����Ⱦ��֮һ���ҹ��涨������SO2�������ó���0.02mg/L����1�������ǽ�ˮ�����ԣ�pHС��5.6����ͳ�ƣ�����ط���������2.1��ʳ��pH=3���������������������Ǵ����е�SO2�͵�����������ǵ���Ҫ��Դ��ú��ʯ�͵�ȼ�գ�ȫ����ÿ���ŷ�1.5�ڶֵ�SO2��
��SO2���ڿ������ܹ��յ��������ö�����������������ˮ�γ����������꣮��д����������Ӧ�Ļ�ѧ����ʽ
������ɵ��µ�Σ����
A����ʴ������ B��������ľ��ή C����ɺ����ֺ� D���������
��Ϊ�˼���������γɣ��������SO2���ŷ�������ȼ���е��������
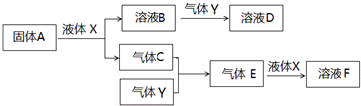
��2��ij��ѧ��ȤС��ѡ����ͼʵ��װ�ã��ⶨ��ҵԭ��������SO2��N2��O2����SO2�ĺ�������֪��������H2SO3������H2SO4����
����ԭ��������������ʱ������װ�����ӵ�˳���ǣ�ԭ������
�ڵ�װ�â��г���
������Ϊ�����Լ��У��������������Թ��еĵ�ĵ�����Һ����
A������KMnO4��Һ�� B��NaOH��Һ C����ˮ D����ˮ
��������Һ��Ũ��Ϊ0.05mol/L�����Ϊ20mL���ռ�����N2��O2�����Ϊ297.6mL��������Ϊ��״���µ����������ô��SO2�ĺ����Ƿꣿ��Ҫ��д�����̣�
���㣺��������Ļ�ѧ����,��ѧ����ʽ���йؼ���,�����������Ⱦ������
ר�⣺������,����Ԫ��
��������1����SO2�����ڿ����о��۳�������ΪSO3��SO3��ˮ��Ӧ��д����Ӧ�Ļ�ѧ����ʽ���ɣ�
�������Σ���ܴ��Ը�ʴ�����������ľ��ή��Ӱ��ũ����������ʹˮ���ữ�ȣ�
�ۼ��ٻ�ʯȼ�ϵ�ȼ�գ�ȼ������Һ���յȴ�ʩ��
��2����ԭ����ͨ����ĵ�����Һ����������H2SO3������H2SO4 �����ն����������壬ͨ��װ�â����ˮ�����ⶨ�����͵����������
��װ�â������ն����������壬��ĵ�����Һ����ɫ������ɫ��ȥ��˵���ⵥ��ȫ����Ӧ��Ӧֹͣͨ����
�۶���������л�ԭ�ԣ��������ø��������Һ���գ����������ԣ����������������ն�������������������������ˮ�������ն��������غ�ɫ��ȥ���������ԣ���ˮ�������ն�������ʵ����������������
�����ݵⵥ��������������Ļ�ѧ����ʽ���������������������õ�ԭ���������������������������������SO2�dz����Ĵ�����Ⱦ��֮һ���ҹ��涨������SO2�������ó���0.02mg/L�����жϣ�
�������Σ���ܴ��Ը�ʴ�����������ľ��ή��Ӱ��ũ����������ʹˮ���ữ�ȣ�
�ۼ��ٻ�ʯȼ�ϵ�ȼ�գ�ȼ������Һ���յȴ�ʩ��
��2����ԭ����ͨ����ĵ�����Һ����������H2SO3������H2SO4 �����ն����������壬ͨ��װ�â����ˮ�����ⶨ�����͵����������
��װ�â������ն����������壬��ĵ�����Һ����ɫ������ɫ��ȥ��˵���ⵥ��ȫ����Ӧ��Ӧֹͣͨ����
�۶���������л�ԭ�ԣ��������ø��������Һ���գ����������ԣ����������������ն�������������������������ˮ�������ն��������غ�ɫ��ȥ���������ԣ���ˮ�������ն�������ʵ����������������
�����ݵⵥ��������������Ļ�ѧ����ʽ���������������������õ�ԭ���������������������������������SO2�dz����Ĵ�����Ⱦ��֮һ���ҹ��涨������SO2�������ó���0.02mg/L�����жϣ�
���
�⣺��1����SO2�����ڿ����о��۳�������ΪSO3��SO3��ˮ��Ӧ���������ᣬ�йط�Ӧ�Ļ�ѧ����ʽΪ��2SO2+O2
2SO3��SO3+H2O�TH2SO4��
�ʴ�Ϊ��2SO2+O2
2SO3��SO3+H2O�TH2SO4��
������ɵ��µ�Σ���У����Ը�ʴ�����������ľ��ή��Ӱ��ũ�����������ʴ�Ϊ��ABD��
��Ϊ�˼���������γɣ����ǿ��Բ�ȡ�Ĵ�ʩ�Ǽ��ٻ�ʯȼ�ϵ�ȼ�յȣ���ȼ���е�����������������Է����еĵ����������ü�Һ���գ������ŷŵ���������Ⱦ������
�ʴ�Ϊ������Һ��
��2����ԭ����ͨ����ĵ�����Һ����������H2SO3������H2SO4 �����ն����������壬ͨ��װ�â����ˮ�����ⶨ�����͵��������������װ�ýӿ�˳��Ϊ��c��d��b��a��e��
�ʴ�Ϊ��c��d��b��a��
��װ�â������ն����������壬��ĵ�����Һ����ɫ������ɫ��ȥ��˵���ⵥ��ȫ����Ӧ��Ӧֹͣͨ������������������ղ���ȫ�������
�ʴ�Ϊ����ɫǡ�ñ����ɫ��
�۶���������л�ԭ�ԣ��������ø��������Һ���գ����������ԣ����������������ն�������������������������ˮ�������ն��������غ�ɫ��ȥ���������ԣ���ˮ�������ն�������ʵ����������������
�ʴ�Ϊ��A��C��
��д������ʽI2+SO2+2H2O=2I-+SO42-+4H+
������֪n��SO2��=n��I2��=0.05 mol/L��0.02 L=0.001 mol����22.4 mL��
���Թ�ҵԭ���������Ϊ 297.6 mL+22.4 mL=320 mL��
����SO2������Ϊ 0.001 mol��64 g/mol=0.064 g��
��SO2�ĺ���Ϊ=200 mg/L��0.02 mg/L��
����SO2�������س��ꣻ
��д������ʽI2+SO2+2H2O=2I-+SO42-+4H+
������֪n��SO2��=n��I2��=0.05 mol/L��0.02 L=0.001 mol����22.4 mL��
�ҵԭ���������Ϊ 297.6 mL+22.4 mL=320 mL��
����SO2������Ϊ 0.001 mol��64 g/mol=0.064 g��
��SO2�ĺ���Ϊ=200 mg/L��0.02 mg/L��
����SO2�������س��ꣻ
| ||
�ʴ�Ϊ��2SO2+O2
| ||
������ɵ��µ�Σ���У����Ը�ʴ�����������ľ��ή��Ӱ��ũ�����������ʴ�Ϊ��ABD��
��Ϊ�˼���������γɣ����ǿ��Բ�ȡ�Ĵ�ʩ�Ǽ��ٻ�ʯȼ�ϵ�ȼ�յȣ���ȼ���е�����������������Է����еĵ����������ü�Һ���գ������ŷŵ���������Ⱦ������
�ʴ�Ϊ������Һ��
��2����ԭ����ͨ����ĵ�����Һ����������H2SO3������H2SO4 �����ն����������壬ͨ��װ�â����ˮ�����ⶨ�����͵��������������װ�ýӿ�˳��Ϊ��c��d��b��a��e��
�ʴ�Ϊ��c��d��b��a��
��װ�â������ն����������壬��ĵ�����Һ����ɫ������ɫ��ȥ��˵���ⵥ��ȫ����Ӧ��Ӧֹͣͨ������������������ղ���ȫ�������
�ʴ�Ϊ����ɫǡ�ñ����ɫ��
�۶���������л�ԭ�ԣ��������ø��������Һ���գ����������ԣ����������������ն�������������������������ˮ�������ն��������غ�ɫ��ȥ���������ԣ���ˮ�������ն�������ʵ����������������
�ʴ�Ϊ��A��C��
��д������ʽI2+SO2+2H2O=2I-+SO42-+4H+
������֪n��SO2��=n��I2��=0.05 mol/L��0.02 L=0.001 mol����22.4 mL��
���Թ�ҵԭ���������Ϊ 297.6 mL+22.4 mL=320 mL��
����SO2������Ϊ 0.001 mol��64 g/mol=0.064 g��
��SO2�ĺ���Ϊ=200 mg/L��0.02 mg/L��
����SO2�������س��ꣻ
��д������ʽI2+SO2+2H2O=2I-+SO42-+4H+
������֪n��SO2��=n��I2��=0.05 mol/L��0.02 L=0.001 mol����22.4 mL��
�ҵԭ���������Ϊ 297.6 mL+22.4 mL=320 mL��
����SO2������Ϊ 0.001 mol��64 g/mol=0.064 g��
��SO2�ĺ���Ϊ=200 mg/L��0.02 mg/L��
����SO2�������س��ꣻ
���������⿼���˶����������ʷ��������������ͼ���Ӧ�ã�ע��ʵ��ⶨ���̵ķ����жϣ����ջ����ǹؼ�����Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
�����Ŀ
����˵������ȷ���ǣ�������
| A�������ױ��ں˴Ź����������ܳ�������壬��������֮��Ϊ3��1 |
B����ϵͳ��������������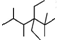 ��������2��2��4��5-�ļ�-3��3-���һ����� ��������2��2��4��5-�ļ�-3��3-���һ����� |
C���ṹƬ��Ϊ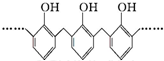 �ĸ߾���䵥���Ǽ�ȩ�ͱ��� �ĸ߾���䵥���Ǽ�ȩ�ͱ��� |
D������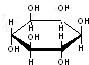 �������� ��������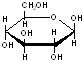 ��Ԫ�������ͬ����ѧʽ��ΪC6H12O6������Cm��H2O��n����ˣ�������������� ��Ԫ�������ͬ����ѧʽ��ΪC6H12O6������Cm��H2O��n����ˣ�������������� |
2014��4��10�գ����ݷ�������ˮ�����������¼�������ʯ�ͻ����Ļ���ԭ�ϣ������������ڣ�������Ѫ��֯�����γ���ѪҺ���ԵĴ�л���������������֯�϶�Ϊ�°����ʣ����й��ڱ���������ȷ���ǣ�������
| A�������ǵ���˫��������ɵ�ƽ�滷״�ṹ | |||
| B��������ϩ����ʹ����KMnO4��Һ��ɫ | |||
C������������Ӧ��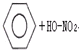
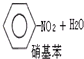 ����ȡ����Ӧ ����ȡ����Ӧ | |||
| D��������ˮ�ڴ��������·���ȡ����Ӧ�����屽 |
���и��������ڲ�ͬ�����·�Ӧʱ�����ܵõ���ͬ���������Ҫ�����ڷ�Ӧ���Ũ�Ȳ�ͬ��������ǣ�������
| A��Fe+H2SO4 |
| B��Cu+HNO3 |
| C��NaOH+CO2 |
| D��AlCl3+NaOH |
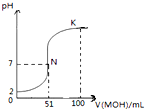 ��1�������£�������һԪ��ֱ��NaOH��Һ�������ϣ�ʵ���������£�
��1�������£�������һԪ��ֱ��NaOH��Һ�������ϣ�ʵ���������£�
 ��
�� E��CH4��CH3��CH2��2CH3
E��CH4��CH3��CH2��2CH3 ��80��ʱ��0.40mol��N2O4�������2L�Ѿ���յĹ̶��ݻ����ܱ������з������·�Ӧ��
��80��ʱ��0.40mol��N2O4�������2L�Ѿ���յĹ̶��ݻ����ܱ������з������·�Ӧ��