��Ŀ����
��ʯ���顢ʯ�ҵ�(CaCN2)������������H2S����Ӧ�����ܾ���β�������ܻ��Ӧ�ù㷺��CS(NH2)2(����)���䲿�ֹ����������£�
��1�������£�H2S�������з�Ӧ:2H2S(g)  2H2(g)+S2(g),��ƽ�ⳣ������ʽΪK=��
2H2(g)+S2(g),��ƽ�ⳣ������ʽΪK=��
��2����ʯ��������H2S��ȡCa(HS)2��Ҫ�ڵ����½��У���ԭ������ �����˵õ��������������ã���������Ҫ�ɷ����� (�ѧʽ)��
��3���ϳ������賤ʱ����裬���ڽϸ��¶�(80��-85��)�½��У���Ŀ������ ��
Ca(HS)2��CaCN2��ˮ��Һ�кϳ������Ļ�ѧ����ʽΪ�� ��
��4��������X�����廥Ϊͬ���칹�壬X����FeCl3��Һ�У���Һ�Ժ�ɫ��X�Ļ�ѧʽΪ�� ��
��1��c2(H2)��c(S2)/c2(H2S)
��2���¶ȵ�H2S�ܽ�������Һ��H2SŨ�ȸߣ����������գ�Ca(OH)2[��Ca(OH)2��CaS]
��3��ʹ��Ӧ���ֽӴ���ά�ֽϸ��¶ȣ���������߷�Ӧ����
Ca(HS)2+2CaCN2+6H2O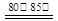 2CS(NH2)2 + 3Ca(OH)2
2CS(NH2)2 + 3Ca(OH)2
��4��NH4SCN
���������������1��K�ı���ʽΪ������Ũ�ȵĴη����Է�Ӧ��Ũ�ȵĴη�����2��������ˮ�е��ܽ�ȣ������¶ȵ����߶���С��ʯ������Ca(OH)2��Ũ�Ƚϴ�����H2S��һ������ʣ�࣬���������á���3���Ӵ�������¶ȸ߾�������߷�Ӧ���ʡ��ɣ�2������ʾ֪��ʯ��������H2S����Ca(HS)2���롰�ϳɡ��Σ���ͷָ��Ϊ���뷴Ӧ���CaCN2��H2O��Ϊ��Ӧ���Ԫ���غ�֪������CS(NH2)2���Ca(OH)2����4����FeCl3��Һ����Һ�Ժ�ɫ��ӦΪSCN����ͬ���칹��Ļ�ѧʽ��ͬ����ԭ���������Ŀ����ȷ������NH4�� ����ΪNH4SCN��
���㣺��ѧ�������̡�Ԫ�ؼ��������ѧ��������

���������LiFePO4��һ��������������ӵ�صĵ缫���ϡ�ij�����������졢﮻�ʯLiAl��SiO3��2��������Ca2+��Mg2+���Σ���̼�۵�ԭ����������������ﮡ�����Ҫ�����������£�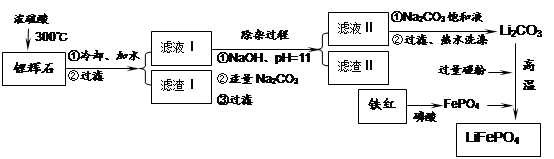
��֪��2LiAl��SiO3��2 + H2SO4(Ũ) 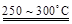 Li2SO4 + Al2O3��4SiO2��H2O��
Li2SO4 + Al2O3��4SiO2��H2O��
| �¶�/�� | 20 | 40 | 60 | 80 |
| �ܽ��(Li2CO3)/g | 1.33 | 1.17 | 1.01 | 0.85 |
| �ܽ��(Li2SO4)/g | 34.2 | 32.8 | 31.9 | 30.7 |
��1�����������пɷ����Al2O3������ͼ��ʾ����д�����ɳ��������ӷ���ʽ ��
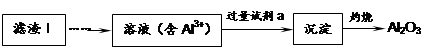
��2�����������Ҫ�ɷ��ǣ� ���ѧʽ����
��3������Һ���м��뱥��Na2CO3��Һ�����˺��á���ˮϴ�ӡ���ԭ����
��
��4��д���ڸ�����������������﮵Ļ�ѧ����ʽ ��
��5����������﮵���ܷ�ӦΪ��FePO4+Li
 LiFePO4������еĹ������ʿɴ���Li������д���õ�طŵ�ʱ��������Ӧ�� �����øõ�ص�ⱥ��ʳ��ˮ�����ص缫��Ϊ���Ե缫������������������4480mL���壨��״��������ʱ���õ������﮵�����Ϊ ��
LiFePO4������еĹ������ʿɴ���Li������д���õ�طŵ�ʱ��������Ӧ�� �����øõ�ص�ⱥ��ʳ��ˮ�����ص缫��Ϊ���Ե缫������������������4480mL���壨��״��������ʱ���õ������﮵�����Ϊ �� �Ե���±ˮ����Ҫ��NaCl����������Ca2+��Mg2+��Ϊ��Ҫԭ�������������Ƶ��¹������£�ͬʱ�ܵõ��������ʵĸ���Ʒ�Ȼ�李�
��֪�����������ʵ��ܽ������ͼ��
��1�������ӡ�ʱ���ȼ�������ʯ������˳�ȥMg2+����ͨ��CO2����������ˮ����pH���˳�ȥCa2+��������������Ҫ�ɷ�Ϊ �� ��
��2��������1���Ļ�ѧʽΪ ��
��3���ڡ���Һ1���м��������Ŀ���� ��������2���Ļ�ѧʽΪ ��
��4����֪H2CO3��H2SO3�ĵ��볣�����±�����ͨ��SO2����Ӧ�Ļ�ѧ����ʽΪ ��
| ���� | ���볣����25�棩 |
| H2CO3 | K1=4.4X10��7 K2=4.7X10��11 |
| H2SO3 | K1=1.23X10��2 K2=5.6X10��8 |
�����Ṥҵ�����У�Ϊ��������SO2��ת�������ܳ���������ܣ��������м����Ƚ������Ӵ��ң���ͼ���������ܱ���ϵ���������������A������������Ϊ( )
| A��SO2 | B��SO3��O2 | C��SO2��SO3 | D��SO2��O2 |
������(AlN)�������¡�������������Ժõ��������ʣ����㷺Ӧ���ڵ��ӹ�ҵ���մɹ�ҵ��������һ�������£���������ͨ�����·�Ӧ�ϳɣ�Al2O3��N2��3C=2AlN��3CO������������ȷ����(����)��
| A���ڵ������ĺϳɷ�Ӧ�У�N2�ǻ�ԭ����Al2O3�������� |
| B��������Ӧ��ÿ����2 mol AlN��N2�õ�3 mol���� |
| C���������е�Ԫ�صĻ��ϼ�Ϊ��3�� |
| D�����������ڸ��ϲ��� |
Ϊ��ͬʱʩ�÷ֱ����ס�������Ԫ�صĻ��ʣ������ṩ�Ļ��ʣ���K2CO3����KCl����Ca(H2PO4)2����(NH4)2SO4���ݰ�ˮ�������˵������(����)
| A���٢ڢۢ� | B���ڢۢ� | C���٢ڢ� | D���ۢܢ� |





