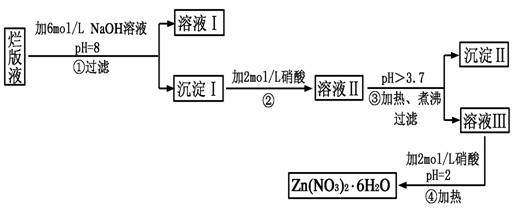
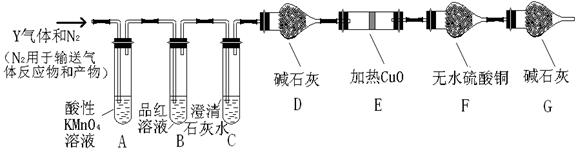
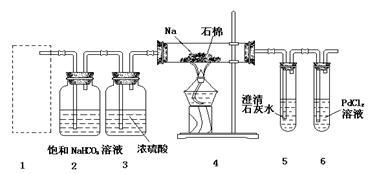
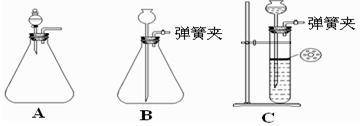
��Ŀ����
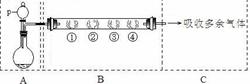
ʵ���ҳ����ü�ȩ��(HCHO)�ⶨ(NH4)2SO4��Ʒ�е��������������䷴Ӧԭ��Ϊ��
4NH4�� ��6HCHO =3H����6H2O��(CH2)6N4H�� �۵ζ�ʱ��1 mol (CH2)6N4H������NaOH�� l mol H���൱�ݣ�Ȼ����NaOH����Һ�ζ���Ӧ���ɵ��ᡣij��ȤС���ü�ȩ������������ʵ�飺
����I ��ȡ��Ʒ1.500 g��
����II ����Ʒ�ܽ����ȫת�Ƶ�250 mL����ƿ�У����ݣ����ҡ�ȡ�
����III ��ȡ25.00 mL��Ʒ��Һ��250 mL��ƿ�У�����10 mL 20�������Լ�ȩ��Һ��ҡ�ȡ�����5 min����1~2�η�̪��Һ����NaOH����Һ�ζ����յ㡣�����������������ظ�2�Ρ�
(1)���ݲ���III��գ�
�ټ�ʽ�ζ���������ˮϴ�Ӻ�ֱ�Ӽ���NaOH����Һ���еζ���������Ʒ�е�����������________(�ƫ�ߡ�����ƫ�͡�����Ӱ�족)��
����ƿ������ˮϴ�Ӻ�ˮδ��������ζ�ʱ��ȥNaOH����Һ�����________________(�ƫ����ƫС������Ӱ�족)
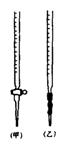
�۵ζ�ʱ�ߵα�ҡ����ƿ���۾�Ӧ�۲�____________
(A)�ζ�����Һ��ı仯 (B)��ƿ����Һ��ɫ�ı仯
�ܵζ��ﵽ�յ�ʱ����ָ̪ʾ����_________ɫ���_________ɫ��
(2)�ζ�������±���ʾ��
�ζ�ʱ����NaOH����Һ�������ƽ��ֵV="__________mL;" ��NaOH����Һ��Ũ��Ϊ0.1010 mol��L��1����250 mL��Һ�е�n(NH4+)=__________mol,����Ʒ�е�����������Ϊ______��
4NH4�� ��6HCHO =3H����6H2O��(CH2)6N4H�� �۵ζ�ʱ��1 mol (CH2)6N4H������NaOH�� l mol H���൱�ݣ�Ȼ����NaOH����Һ�ζ���Ӧ���ɵ��ᡣij��ȤС���ü�ȩ������������ʵ�飺
����I ��ȡ��Ʒ1.500 g��
����II ����Ʒ�ܽ����ȫת�Ƶ�250 mL����ƿ�У����ݣ����ҡ�ȡ�
����III ��ȡ25.00 mL��Ʒ��Һ��250 mL��ƿ�У�����10 mL 20�������Լ�ȩ��Һ��ҡ�ȡ�����5 min����1~2�η�̪��Һ����NaOH����Һ�ζ����յ㡣�����������������ظ�2�Ρ�
(1)���ݲ���III��գ�
�ټ�ʽ�ζ���������ˮϴ�Ӻ�ֱ�Ӽ���NaOH����Һ���еζ���������Ʒ�е�����������________(�ƫ�ߡ�����ƫ�͡�����Ӱ�족)��
����ƿ������ˮϴ�Ӻ�ˮδ��������ζ�ʱ��ȥNaOH����Һ�����________________(�ƫ����ƫС������Ӱ�족)
�۵ζ�ʱ�ߵα�ҡ����ƿ���۾�Ӧ�۲�____________
(A)�ζ�����Һ��ı仯 (B)��ƿ����Һ��ɫ�ı仯
�ܵζ��ﵽ�յ�ʱ����ָ̪ʾ����_________ɫ���_________ɫ��
(2)�ζ�������±���ʾ��
| �ζ� ���� | ������Һ����� /mL | ����Һ�����/mL | |
| �ζ�ǰ�̶� | �ζ���̶� | ||
| 1 | 25.00 | 1.02 | 21.03 |
| 2 | 25.00 | 2.00 | 21.99 |
| 3 | 25.00 | 0.20 | 20.20 |
(1)��ƫ�ߣ�2�֣��� ����Ӱ�죨2�֣� ��B��2�֣��� ���ޣ�1�֣��� �ۺ�(��dz��) ��1�֣��� (2) 20.00 ��1�֣� 0.0202 ��1�֣� 18.85%����1�֣�
��

��ϰ��ϵ�д�
 ������ѧ���̲���ȫ���ϵ�д�
������ѧ���̲���ȫ���ϵ�д� ������ʱ����ҵ����ϵ�д�
������ʱ����ҵ����ϵ�д� ��ĩ���ƾ�ϵ�д�
��ĩ���ƾ�ϵ�д� ���ɿ��ñ���ϵ�д�
���ɿ��ñ���ϵ�д�
�����Ŀ
 a2CO3��NaHCO3��MnO2��Na2O2��NaCl����ˮCaCl2��NH4HCO3����ʯ�ҵȹ����H2O2������ˮ��
a2CO3��NaHCO3��MnO2��Na2O2��NaCl����ˮCaCl2��NH4HCO3����ʯ�ҵȹ����H2O2������ˮ��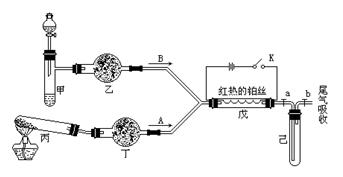
 ��
��