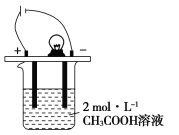
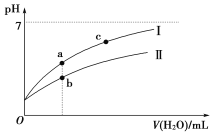
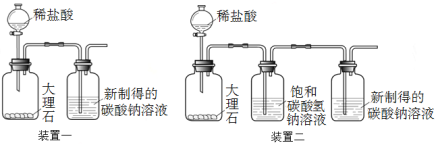
题目内容
【题目】听下面一段对话,完成五道小题,每小题仅填写一个词。这段对话你将听两遍。
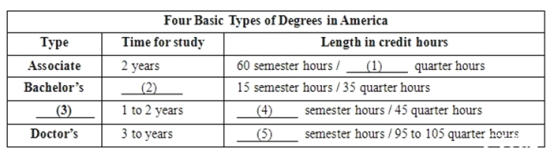
【答案】
(1) 95
(2) 4
(3) Master's
(4) 30
(5) 62 to 72
【解析】听力原文:
There are four basic types of college degrees in America, starting with the associate degree. The associate takes about two years to complete when one is enrolled full-time. The bachelor's degree takes four years of full-time study with the master's taking one to two years, and the doctor's three to four years. The associate degree may be substituted for the first two years of a bachelor's degree if it is a transfer degree. Not all associate degrees are designed for a transfer. Some are technical degrees called terminal degrees, which means they do not count toward a bachelor's. The bachelor's is normally required before one can work at the master's level. Likewise, the master's is normally required before one can work at the doctor's level.
The length in credit hours of university degree programs varies from one school to another. In general, the AA is 60 semester hours or 95 quarter hours. The BA (or BS) is 15 semester hours or 35 quarter hours. The MA is 30 semester hours or 45 quarter hours, and the Ph. D. is 60 to 72 semester hours or 95 to 105 quarter hours.

 智能训练练测考系列答案
智能训练练测考系列答案 计算高手系列答案
计算高手系列答案