��Ŀ����
����Ŀ����������������������BaCl2��ų��ж�����H2S(��ˮ��Һ������)������NaOH����H2S��ȡNa2S���д������±�ΪH2S���չ��յĸ��������ɱ���
��Ŀ | ����(Ԫ/��) |
NaOH | 6000 |
Na2S | 5000 |
��������(�Դ���1��H2S��) | 1114 |
��1����ҵ����β���迼�Ǿ���Ч������Ч�棬�����������Ϸ��������Ӿ���Ч�濼�ǣ����ִ���H2S�ķ�����______________(����ӯ��������������)�ģ�
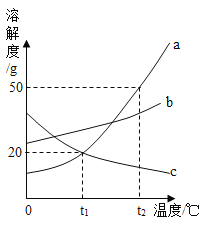
��2����������������������BaCl2��Һ�г���������ᣬΪ�˲ⶨ����Һ��BaCl2����������������ijͬѧ��������ʵ�飺ȡ�ò�ƷҺ10.0 g���ձ��У���ε�����������Ϊ5.3%��Na2CO3��Һ����������¼����Na2CO3��Һ���������й����ı仯��ϵ��ͼ1��ͼ2��ʾ��
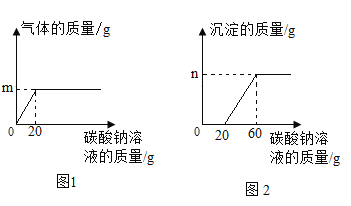
��m��________��n��________��
�����Ȼ�����Ӧ��̼������Һ��������________g��
����������Һ��BaCl2������������(д���������)
���𰸡�(1)����(2) ��0.44��3.94����40����41.6%���������)
��������
��1���ɻ�ѧ����ʽ��֪��2NaOH��H2S=Na2S��2H2O��ÿ80�ݵ�����������ȫ��Ӧ�ܲ���78�ݵ����ƣ������������Ƶ��������������Ƶ���������ȣ����������Ƶĵ���Ϊ6000Ԫ/�֣�����Ҫ��������1114Ԫ/�֣����ɵ����Ƶĵ���Ϊ5000Ԫ/�֣����������ɱ����ڲ�Ʒ�ɱ������ִ���H2S�ķ����ǿ���ġ�
��2�� ���裺����������̼������Ϊm

![]() m=0.44g
m=0.44g
����ͼ��֪����̼������Һ�����ӵ�20 gʱ����Һ������ǡ�÷�Ӧ�ꡣ��BaCl2��Ӧ��̼������Һ������Ϊ60 g��20 g��40 g����10.0 g����Һ���Ȼ���������Ϊx������̼�ᱵ������Ϊn��
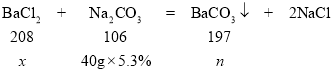
![]() x��4.16 g
x��4.16 g
![]() n=3.94g
n=3.94g
����Һ���Ȼ�������������=![]() ��100%��41.6%
��100%��41.6%

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�����Ŀ�����и�ѡ������ͼ��ʾ�����������
ѡ�� | ʵ����� | ������ | ������ |
A | ��һ��������п���м���ϡ���� | ����ϡ���������/g | ��������������/g |
B | ��һ����������������Һ�м���ͭ�� | ����ͭ�۵�����/g | ��Һ������/g |
C | ��һ��������ϡ�����в��ϼ���̼��Ʒ�ĩ | ����̼��Ƶ�����/g | ��Һ������/g |
D | ���Ȼ��Ʊ�����Һ�м���ˮ | ����ˮ������/g | ���ʵ�����/g |
A.AB.BC.CD.D