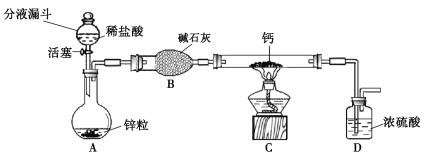
��Ŀ����
����Ŀ����λͬѧ������ʦ����ʵ����ʱ���ڱ�������������ҩ���з���һƿʧȥ��ǩ�ĺ�ɫ��ĩA���ڱ���������������ҩ���з�����һƿʧȥ��ǩ�ĺ�ɫ��ĩB��
��������⣩����ƿ��ɫ��ĩA��B�ֱ���ʲô���ʣ�
�����룩
��ͬѧ��A�����ۣ�B�Ƕ������̣�
��ͬѧ��A��̿�ۣ�B������ͭ��
��ʵ��̽����
ʵ�鲽�� | ʵ������ | ʵ����� |
��ȡ����A��ĩ���Թ��У���������ϡ���ᣮ | ��ɫ��ĩ���ܽ⣬û����������� | A��ĩһ������_____�� |
��ȡ����B��ĩ���Թ��У���������ϡ���ᣮ | _____����Һ����ɫ�� | B��ĩ������ͭ�� |
����˼����չ��
��1���������ֺ�ɫ��ĩ��ϼ��ȣ��۲쵽��ɫ��ĩ��Ϊ��ɫ������ʯ��ˮ����ǣ���ɫ��ĩA��B������Ӧ�Ļ�ѧ����ʽΪ_____��
��2����ͬѧ�IJ�����ȷ��
���𰸡����� ��ɫ��ĩ�ܽ� 2CuO+C![]() 2Cu+CO2��
2Cu+CO2��
��������
{ʵ��̽��}�ٺ�ɫ��ĩ���ܽ⣬û�������������һ���������ۣ���Ϊ�����ܺ�ϡ���ᷴӦ�����������������ۣ�
������ͭ��ϡ���ᷴӦ��������ͭ��ˮ����۲쵽�������Ǻ�ɫ��ĩ�ܽ⣬��Һ����ɫ�������ɫ��ĩ�ܽ⣻
{��˼��չ}��1�������Ŀ�֪���ڸ��������£�̼������ͭ��Ӧ�����˶�����̼��ͭ�������ֺ�ɫ��ĩ������Ӧ�Ļ�ѧ����ʽдΪ��2CuO+C![]() 2Cu+CO2����
2Cu+CO2����

 Сѧѧϰ�ð���ϵ�д�
Сѧѧϰ�ð���ϵ�д� Сѧͬ�����������ܾ�ϵ�д�
Сѧͬ�����������ܾ�ϵ�д�����Ŀ����6�֣���ʡ��������ú̿��Դ�ḻ����˹������ú�㼰��Χ�Ҳ��У��Ǿ����к�������ܳƣ���Ҫ�ɷ��Ǽ��顣
��1������д��������ȫȼ�յĻ�ѧ����ʽ��__________________________________��
��2��ú����˹��ը��������Ҫ������
����˹�����ڱ�ը���ķ�Χ�ڣ���________________________________________��
��3�������dz�����������ı�ը���ޣ�����ݴ��жϣ�
����������ը��������____��
��ȼ���� | ��ը���ޣ���������� |
H2 | 4.0%~74.2% |
CH4 | 5%~15% |
CO | 12.5%~74.2% |
��4������ͼ���У���ȼ�պͱ�ը�ص���
![]()
A B C D
��5��ij�ִ����Ŀ����ˡ���Ũ����˹���缼��������Ч����˿������ܱߵ����������õ硣���ּȲ�ú�ַ��������������ŵ���___________________________________��