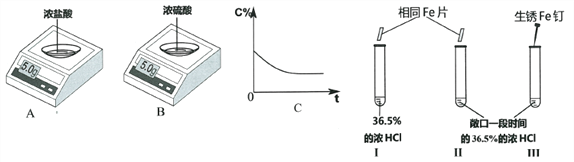
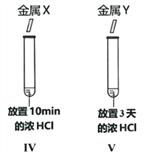
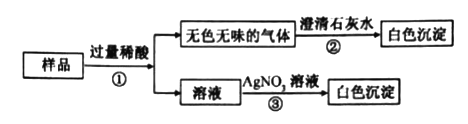
��Ŀ����
����Ŀ���ձ�����һЩ���õ�����������Һ A��Ϊȷ��������������������ʵ����
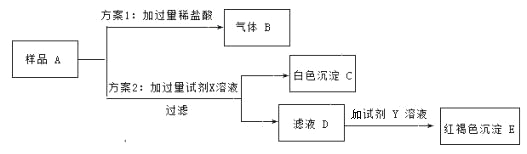
��1���������� B �Ļ�ѧ����ʽ______________��
��2���Լ� X ��______________��ѡ����ᱵ������������������Һ��
��3���������Լ� Y ���Ȼ�����Һ�������ɺ��ɫ���� E �Ļ�ѧ����ʽ______________ ��
��4����Һ D �����ʵijɷ���______________��
��5�����ۣ��ɷ��� 2 ��֪���ձ�������������Һ��______________������ֱ��ʡ�����ȫ���ʡ�����
���𰸡� Na2CO3+2HCl=2NaCl+H2O+CO2�� ���ᱵ��Ba(NO3)2 3NaOH��FeCl3��Fe�vOH�w3����3NaCl �������ƣ������������������ƣ��������� ���ֱ���
��������������ѧ֪ʶ��������Ϣ֪�����õ�����������Һ A�����ܲ��ֱ���������̼���ƣ�̼������ϡ���ᷴӦ�����˶�����̼���壬B�Ƕ�����̼��E�Ǻ��ɫ������Ӧ������������̼�������Ȼ��������ᱵ��Ӧ����̼�ᱵ��ɫ������x���Ȼ��������ᱵ��Y���Ȼ�������1���������� B �Ļ�ѧ����ʽ��Na2CO3+2HCl=2NaCl+H2O+CO2������2���Լ� X �����ᱵ��Һ����3���������Լ� Y ���Ȼ�����Һ�������ɺ��ɫ���� E �Ļ�ѧ����ʽ��3NaOH��FeCl3 ��Fe�vOH�w3 ����3NaCl����4�����������غ㶨��֪����Һ D �����ʵijɷ����������ƣ������������������ƣ��������ơ���5���ɷ��� 2 ��֪���ձ�������������Һ�Dz��ֱ��ʡ�
�㾦�����������Ǻ��ɫ��������ѧ����ʽ����дӦ��ѭ���Կ���ʵΪ��������ѭ�����غ㶨�ɡ�

����Ŀ���Ȼ��ƺ�����þ���ܽ�������������¶��µ��ܽ�ȱ����£�
�¶�/�� | 20 | 30 | 40 | 60 | 80 | 90 | 100 | |
�ܽ�� (g/100GH20) | NaCl | 36.0 | 36.3 | 36.6 | 37.3 | 38.4 | 39.0 | 39.8 |
MgSO4 | 33.7 | 38.9 | 44.5 | 54.6 | 55.8 | 52.9 | 50.4 | |
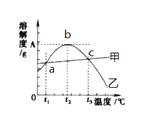
��ش��������⣺
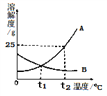
(1)80��ʱ������þ���ܽ��Ϊ__________���ܽ��������������������__________��a���Ӧ���¶�t1��Χ��____________________��
(2)t2��ʱ�к��н϶�NaCl ��MgSO4��Һ��Ϊ�˵õ�������MgSO4���ɲ��õķ���__________
(3)40��ʱ��20g NaCl����50gˮ�У��γɵ���Һ��������������Ϊ__________(��ȷ��0.1%)��Ҫʹt3�汥�͵�MgSO4��Һ�����������������ɲ��õĴ�ʩ��__________��